
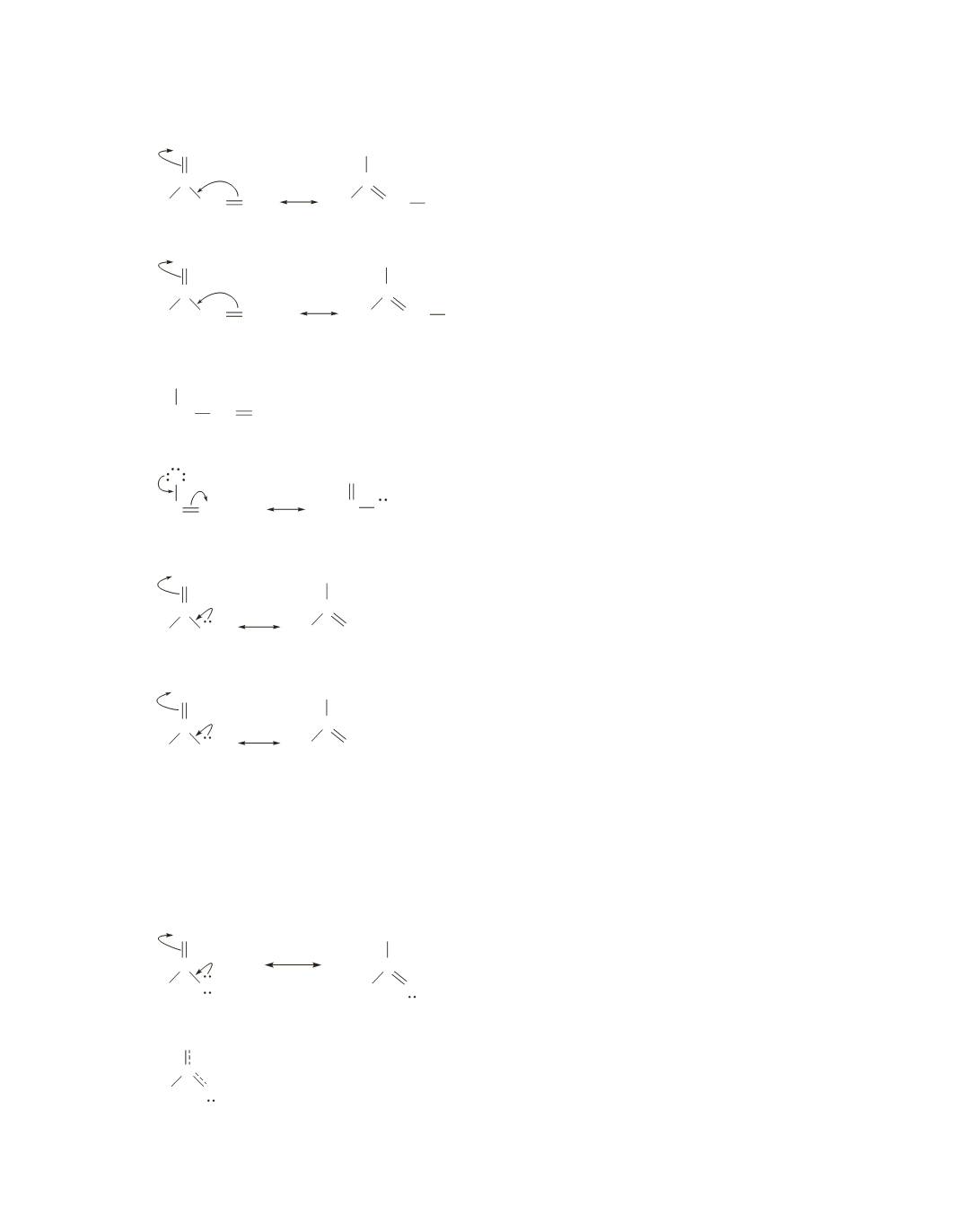
266 Chapter 8
Copyright © 2017 Pearson Education, Inc.
b.
Less stable, because the positive charge is on a primary allylic carbon.
CH
2
CH
2
+
CH
3
C
O
CH
CH
3
C
O
CH
_
CHCH
3
CHCH
3
+
CH
3
C
O
CH
CH
3
C
O
CH
_
More stable, because the positive charge is on a secondary allylic carbon.
c.
CH
3
CH CH CH
2
CH
3
C CHCH
3
O
O
−
CH
3
C CHCH
3
O
−
−
Less stable, because the negative charge is not delocalized.
More stable, because the negative charge is delocalized.
d.
Less stable, because the positive charge is shared by an O and an N.
CH
3
C
NH
2
OH
+
CH
3
C
NH
2
+
OH
CH
3
C
NH
2
CH
3
C
NH
2
+
+
NH
2
NH
2
More stable, because the positive charge is shared by two nitrogens. Nitrogen is less electronegative
than oxygen, so nitrogen is more comfortable with a positive charge.
5.
Solved in the text.
6.
The resonance contributor that makes the greatest contribution to the hybrid is labeled
A
.
B
contributes less to the hybrid than
A
, and
C
contributes less to the hybrid than
B
.
a.
CH
3
C
O
OCH
A
B
3
CH
3
C
O
OCH
3
_
+
A
is more stable than
B
because
B
has
separated charges and has a positive
charge on an oxygen.
CH
3
C
O
OCH
3
δ
–
δ +
















