
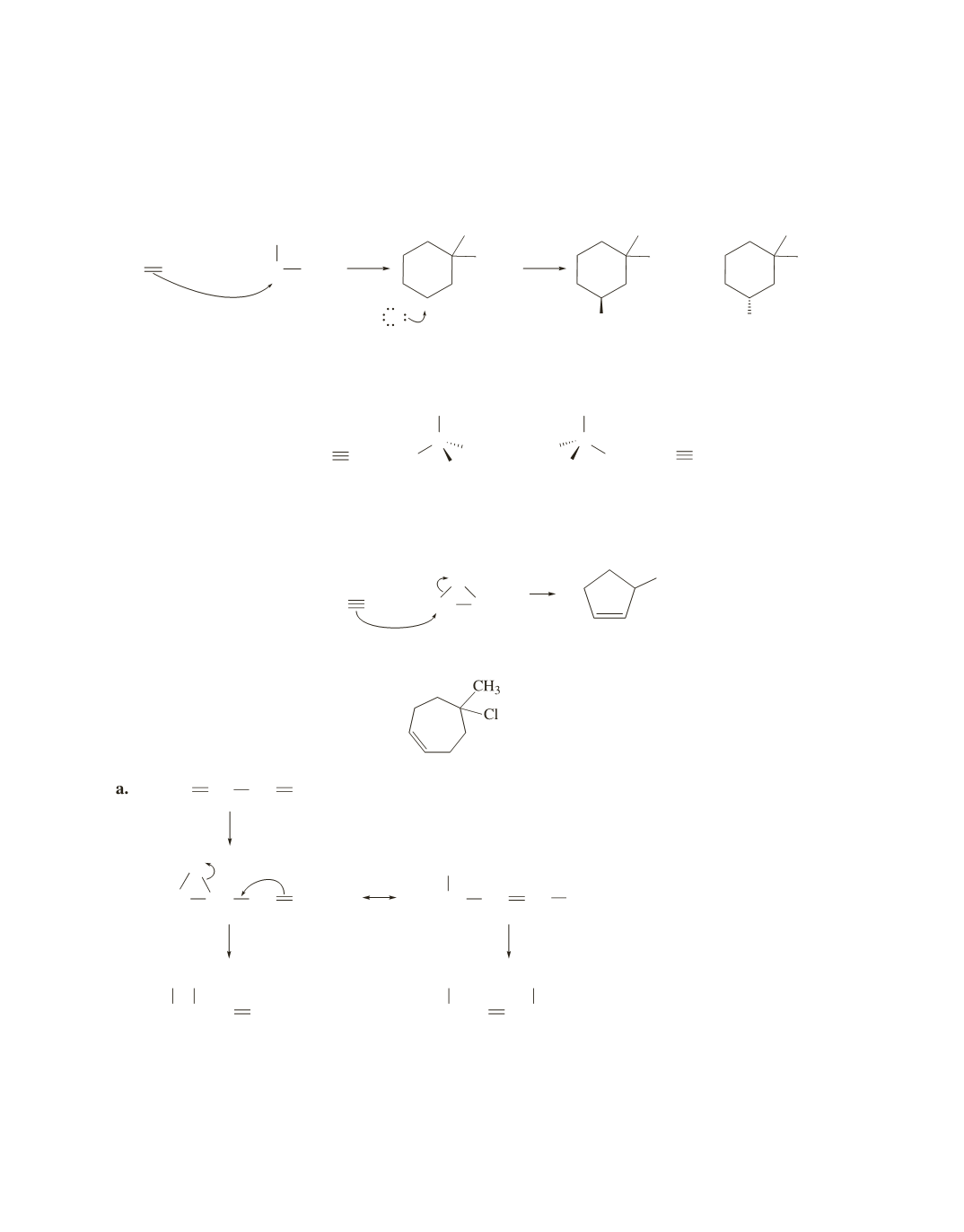
272 Chapter 8
Copyright © 2017 Pearson Education, Inc.
Instead of Br
-
being the nucleophile that adds to the tertiary carbon, the
p
bond can be the nucleophile.
In that case, a stable six-membered ring is formed. (See the Problem-Solving Strategy on page 257 of
the text.) This is expected to be a minor product because, unlike the above reaction of the carbocation with
Br
-
, bond breaking is required to form the product.
CH
2
CH
3
+
CHCH
2
CH
2
CH
2
C CH
3
CH
3
CH
3
Br
− +
CH
3
CH
3
Br
CH
3
CH
3
Br
+
b.
The double bond is more reactive than the triple bond. The reaction forms a new asymmetric center, so
a pair of enantiomers is formed.
CH
2
Cl
C
CCH
2
CH
2
H
Cl
HC
+
CH
2
Cl
C
CH
2
CH
2
C
H
Cl
CH
S
R
+
CH
2
Cl
CCH
2
CH
2
CH
HC
CH
2
Cl
+
It is unlikely that the triple bond will act as a nucleophile, because it would have to form an unstable
vinylic cation intermediate.
c.
The more reactive double bond is the one that forms a tertiary carbocation.
28.
CH
3
CH CH CH CHCH
3
CH
3
CH CH CH CHCH
3
Cl
+
CH
3
CH CH CH CHCH
3
Cl
+
Cl
−
Cl
−
CH
3
CHCHCH CHCH
3
Cl Cl
CH
3
CHCH CHCHCH
3
1,2-addition product
1,4-addition product
Cl
Cl
2
CH
3
CH
2
C C
Br
CH
3
CH
3
CHCH
3
1,2-addition product
+
+
Cl
CH
3
CH
2
C CCHCH
3
Br
CH
3
CH
3
1,4-addition product
















