
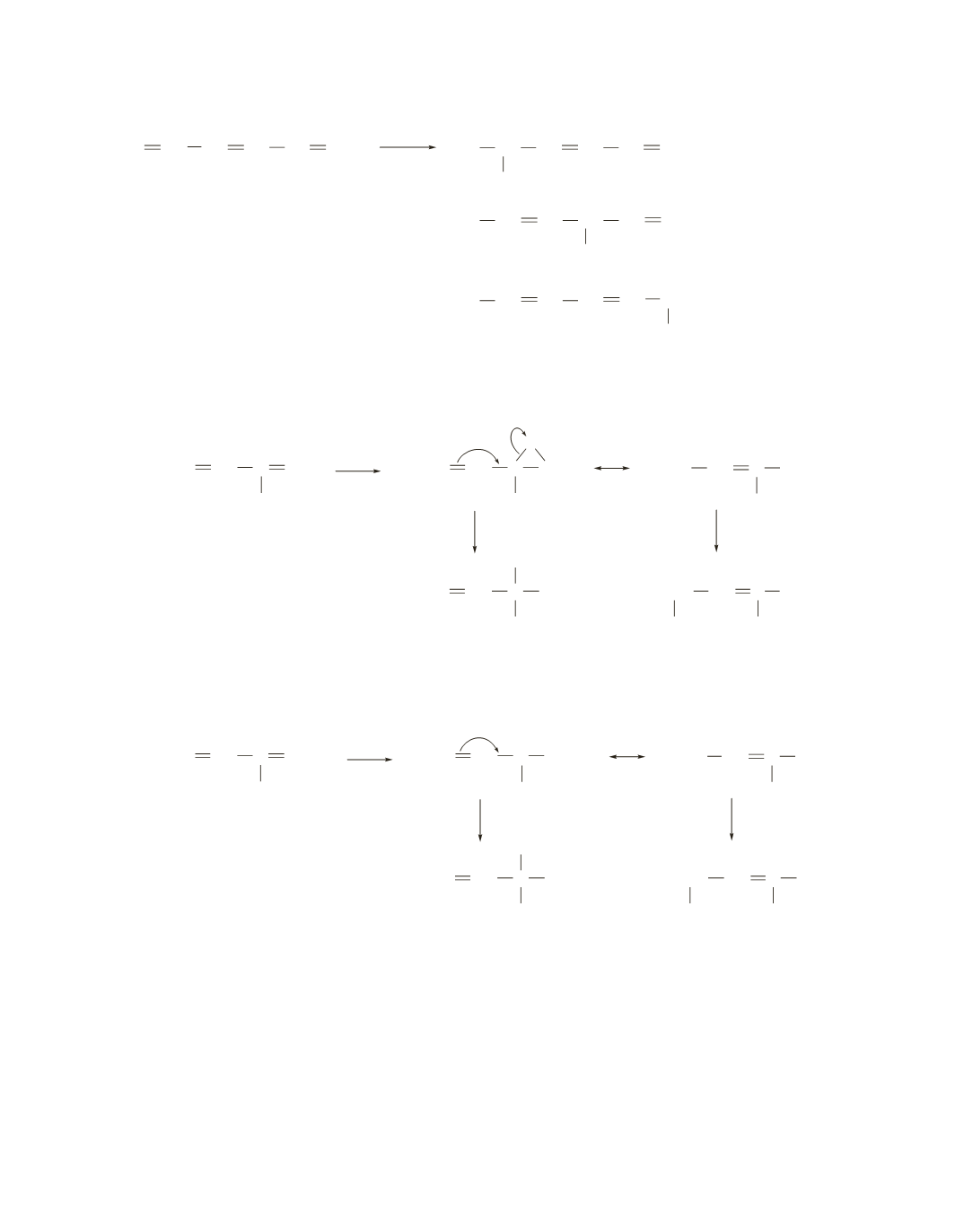
274 Chapter 8
Copyright © 2017 Pearson Education, Inc.
31.
CH
2
CH CH CH CH CH
2
CH
3
CH CH CH CH CH
2
CH
3
CH CH CH CH CH
2
CH
3
CH CH CH CH CH
2
Br
Br
Br
HBr
32.
a.
The chlorine adds so that the positive charge in the resonance contributor is on a secondary allylic
carbon. (If the chlorine had added to the other double bond, the positive charge would be on a primary
allylic carbon.)
CH
3
CH CH C
CH
3
CH
2
CH
3
CH CH C
CH
3
CH
Cl
2
CH
3
CH CH C
CH
3
CH Cl
2
+
+
Cl
−
Cl
−
CH
3
CH CH C CH Cl
2
Cl
CH
3
CH
3
CH CH C
CH
3
CH Cl
2
Cl
Cl
2
b.
The proton adds so that the positive charge in the carbocation is shared by a tertiary allylic and a sec-
ondary allylic carbon. (If the proton had added to the other double bond, the positive charge would be
shared by two secondary allylic carbons.)
CH
3
CH CH C
CH
3
CHCH
3
HBr
CH
3
CH CH C
CH
3
CH
2
CH
3
+
CH
3
CH CH C
CH
3
CH
2
CH
3
+
Br
−
Br
−
CH
3
CH CH C CH
2
CH
3
Br
CH
3
CH
3
CH CH C
CH
3
CH
2
CH
3
Br
33.
a.
Addition at C-1 forms the more stable carbocation because the positive charge is shared by two sec-
ondary allylic carbons. If the deuterium had added to C-4, the positive charge would be shared by a
secondary allylic and a primary allylic carbon.
b.
DCl was used to cause the 1,2- and 1,4-products to be different. If HCl had been used, the 1,2- and
1,4-products would be the same.
















