
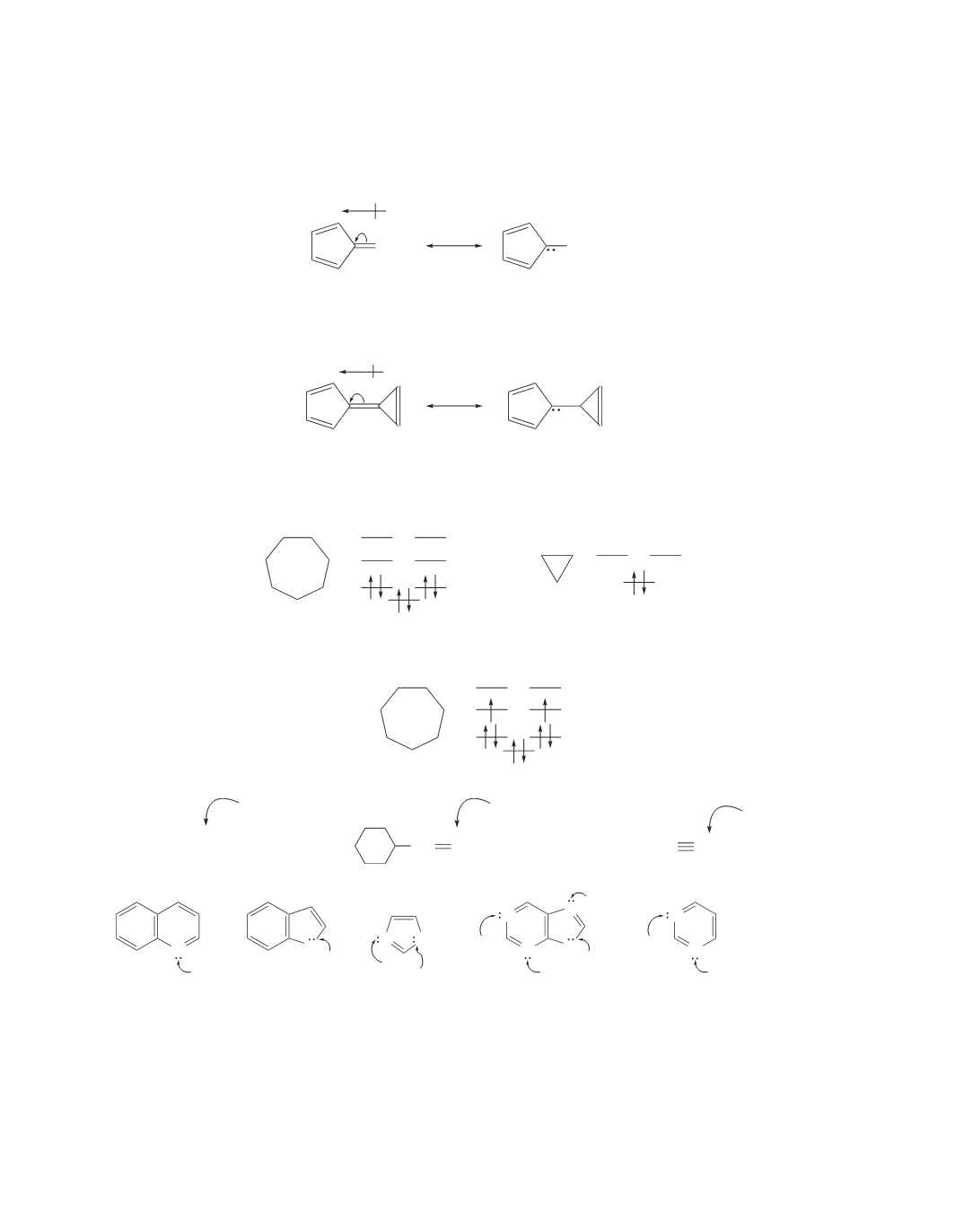
280 Chapter 8
Copyright © 2017 Pearson Education, Inc.
54.
a.
In fulvene, the electrons in the exocyclic double bond (a double bond attached to a ring) move toward
the five-membered ring, because that results in a resonance contributor that is aromatic. Moving the
electrons in the other direction would result in a resonance contributor with an antiaromatic ring.
CH
2
CH
2
fulvene
+
−
b.
In calicene, the electrons in the double bond between the two rings move toward the five-membered
ring, because that results in a resonance contributor with two aromatic rings.
calicene
+−
55.
The Frost circles show that species with completely filled bonding MOs and with electrons in no other
orbitals are aromatic (for example, the cycloheptatrienyl cation and the cyclopropenyl cation).
cycloheptatrienyl cation
cyclopropenyl cation
The species with unpaired electrons in degenerate MOs is antiaromatic (the cycloheptatrienyl anion).
cycloheptatrienyl anion
56.
a.
..
CH
3
CH
2
NH
2
sp
3
b.
CH NCH
2
CH
3
..
sp
2
c.
..
CH
3
CH
2
C N
sp
57.
N
H
p
sp
2
NH N
p
N
sp
2
N
N N
H
N
p
sp
2
sp
2
sp
2
N
N
sp
2
sp
2
58.
Solved in the text.
59.
The lone-pair electrons on both nitrogens in the second compound are
sp
2
electrons; therefore, they are not
part of the
p
cloud and can be protonated without destroying the compound’s aromaticity.
The lone-pair electrons on the nitrogen in the first compound are
p
electrons and are part of the
p
cloud.
If these
p
electrons are protonated, they will no longer be part of the
p
cloud (they cannot be delocalized)
and the compounds will not be aromatic.
















