
Chapter 8 285
Copyright © 2017 Pearson Education, Inc.
14.
minor
minor
major
_
C
CH H
CH CH
2
_
C
CH H
CH CH
2
_
C
CH H
CH CH
2
O
O
O
The major contributor has a negative charge on oxygen, which is more stable than a contributor with a
negative charge on carbon.
15.
Notice that the electrons on the center carbon can be delocalized onto both of the carbonyl oxygens.
_
CH
3
CH
CH
3
_
CH
3
CH
CH
3
major
minor
C
O
C
O
C
O
C
O
_
CH
3
CH
CH
3
_
CH
3
CH
CH
3
major
C
O
C
O
C
O
C
O
b. 2, 4,
and
7
have resonance contributors that all contribute equally to the hybrid.
69.
+
a.
+
b.
c.
−
d.
It is aromatic.
It is aromatic.
It is aromatic.
It is not antiaromatic.
70.
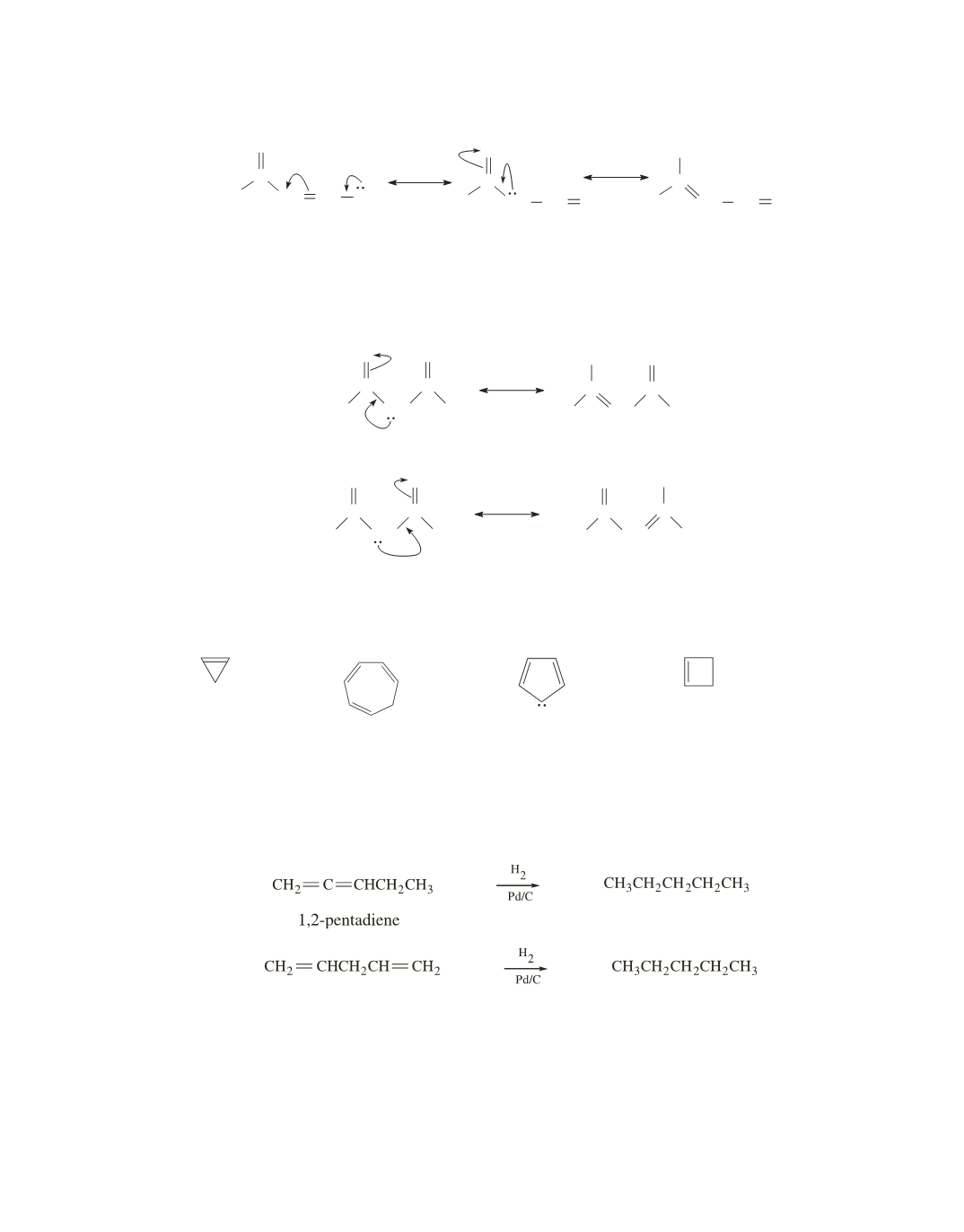
Both compounds form the same product when they are hydrogenated, so the difference in the heats of hy-
drogenation depend only on the difference in the stabilities of the reactants. Because 1,2-pentadiene has
cumulated double bonds and 1,4-pentadiene has isolated double bonds, 1,2-pentadiene is less stable and,
therefore, has a greater heat of hydrogenation (a more negative
∆
H
°
).
1,4-pentadiene
















