
Chapter 8 291
Copyright © 2017 Pearson Education, Inc.
88.
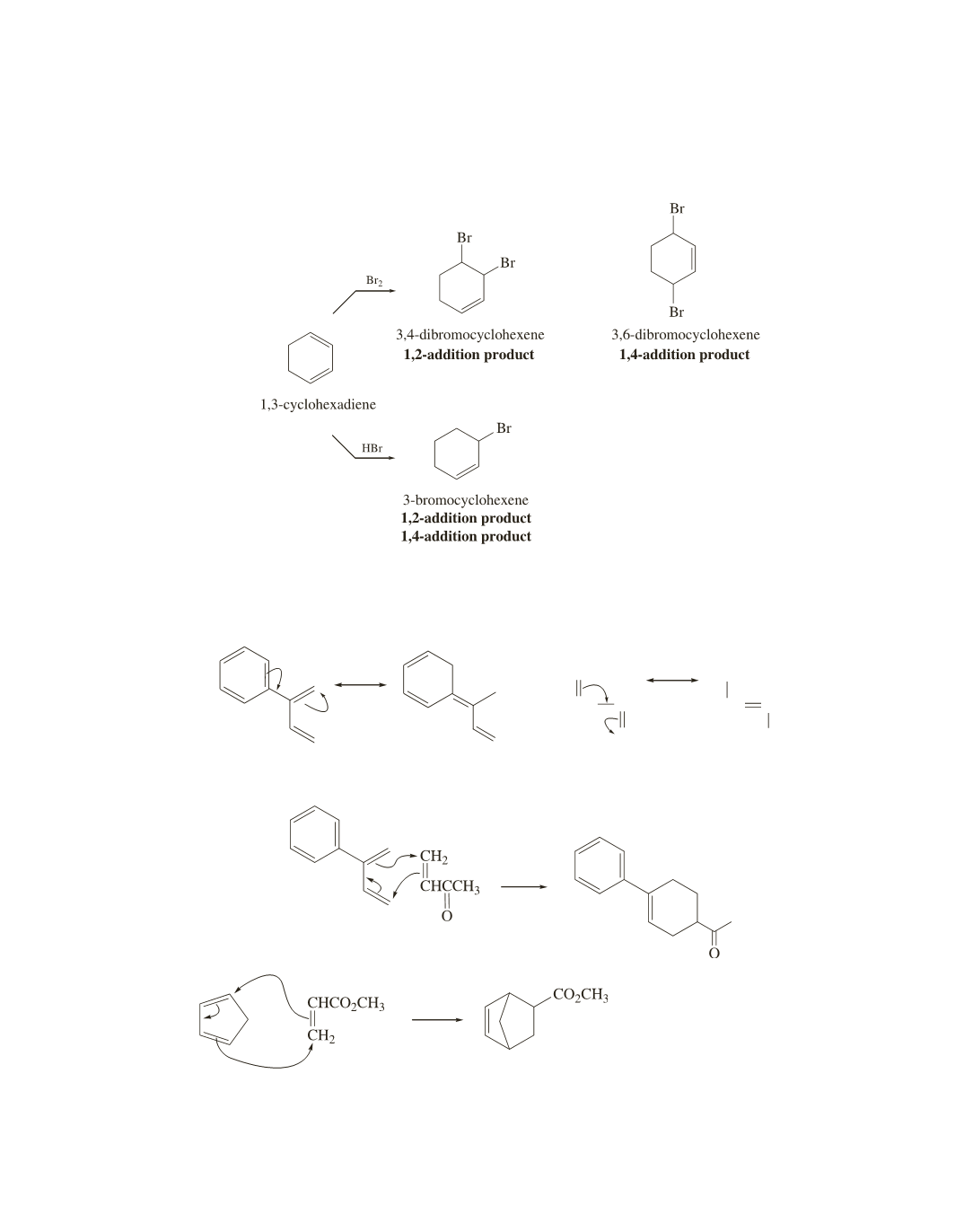
The reaction of 1,3-cyclohexadiene with Br
2
forms 3,4-dibromocyclohexene as the 1,2-addition product
and 3,6-dibromocyclohexene as the 1,4-addition product. The reaction of 1,3-cyclohexadiene with HBr
forms only 3-bromocyclohexene, so it is both the 1,2-addition product and the 1,4-addition product.
+
89.
a.
Only part
a
involves the reaction of two unsymmetrically substituted reactants. Therefore, only for
part
a
do we need to look at the charge distribution in the reactants to determine the major product of
the reaction.
CH
2
CH CCH
3
O
+
_
CH
2
CH CCH
3
O
+
_
We now need to join the negative end of the diene and the positive end of the dienophile.
b.
+
















