
296 Chapter 8
Copyright © 2017 Pearson Education, Inc.
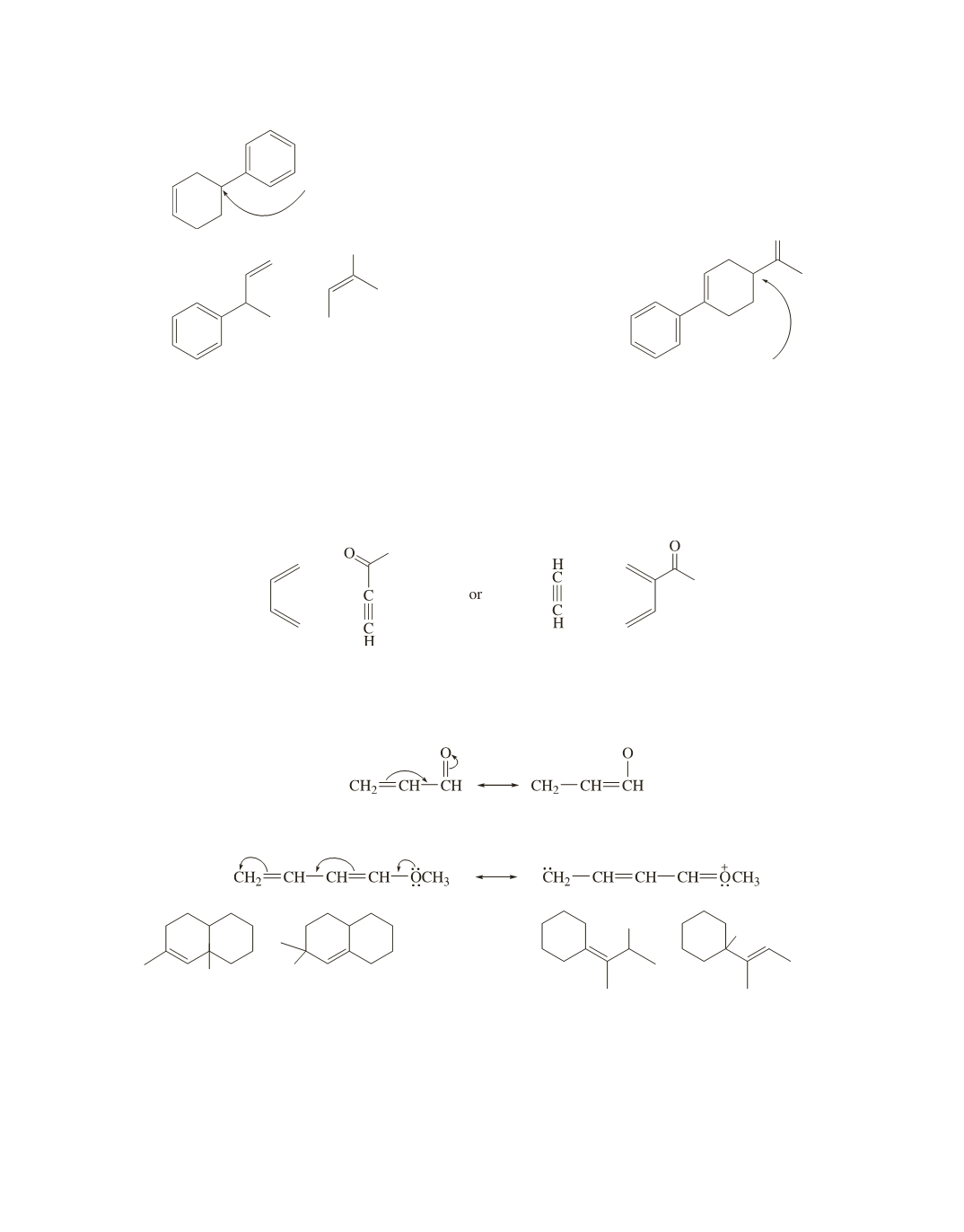
96.
a.
Because the reaction creates an asymmetric center in the product,
the product will be a racemic mixture.
b.
O
−
+
+
−
Even though both reactants are unsymmetrically
substituted, they will be aligned primarily as
shown, because of the relatively stable tertiary
benzylic cation and delocalization of the
p
electrons of the dienophile onto the oxygen.
O
Because the reaction creates an
asymmetric center, the product
will be a racemic mixture.
97.
The first pair is the preferred set of reagents because it has the more nucleophilic diene and the more
electrophilic dienophile.
+
+
98.
A Diels–Alder reaction is a reaction between a nucleophilic diene and an electrophilic dienophile.
a.
The compound shown below is more reactive in both
1
and
2
, because electron delocalization in-
creases the electrophilicity of the dienophile.
−
+
b.
The compound shown below is more reactive, because electron delocalization increases the
nucleophilicity of the diene.
−
99.
a.
Br
1,2-product
kinetic product
1,4-product
Br
b.
Br
Br
1,2-product
kinetic product
thermodynamic product
1,4-product
Both have the same stability,
so neither is the thermodynamic product.
When the reaction is under thernodynamic control,
equal amouts of the two products will be obtained.
















