
300 Chapter 8
Copyright © 2017 Pearson Education, Inc.
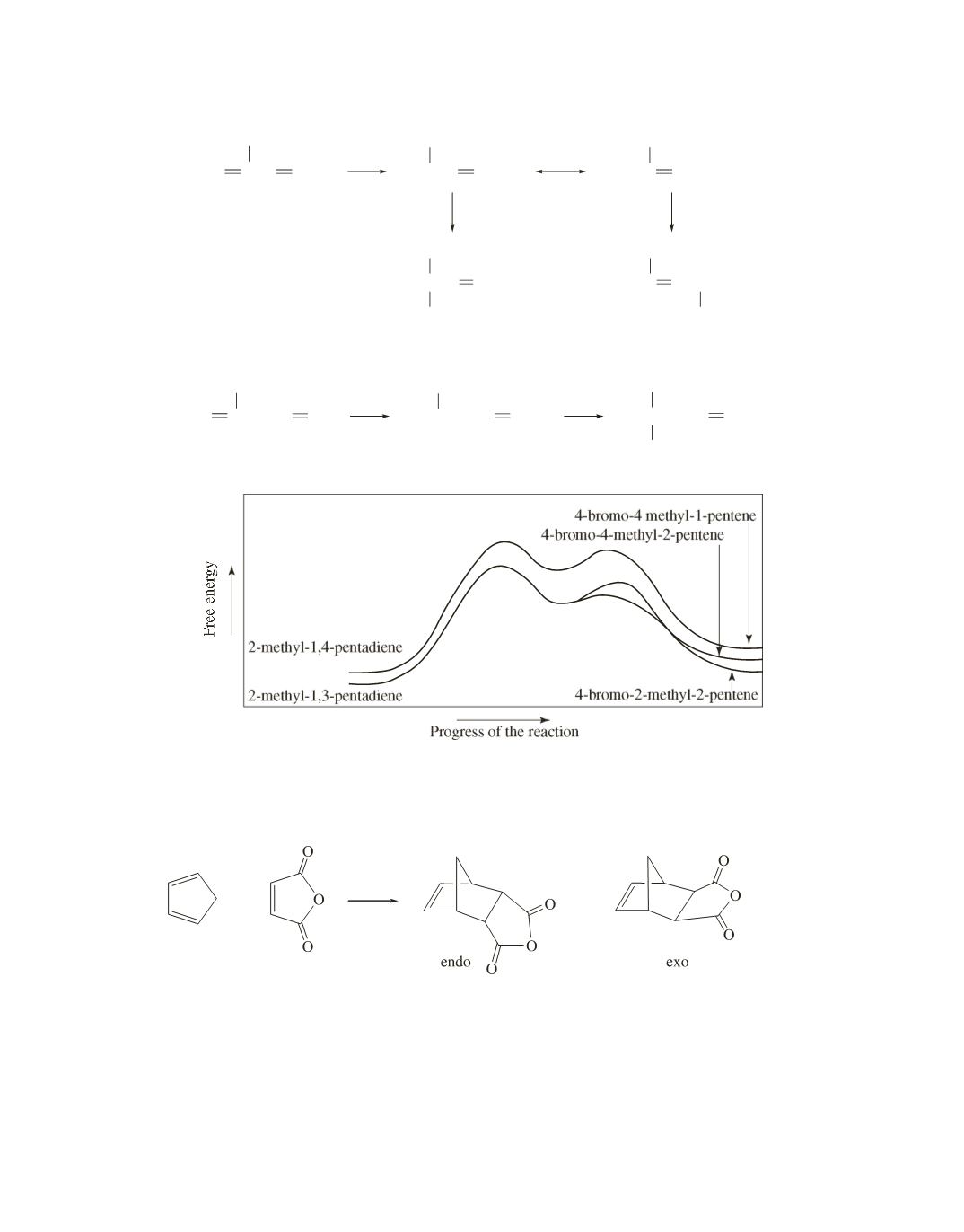
CH
2
CCH CHCH
3
HBr
CH
3
CCH CHCH
3
+
CH
3
Br
−
CH
3
C CHCHCH
3
CH
3
+
CH
3
CCH CHCH
3
CH
3
Br
4-bromo-4-methyl-2-pentene
Br
−
CH
3
C CHCHCH
3
CH
3
4-bromo-2-methyl-2-pentene
Br
CH
3
2-methyl-1,3-pentadiene
CH
2
CCH
2
CH CH
2
HBr
CH
3
2-methyl-1,4-pentadiene
CH
3
CCH
2
CH CH
2
+
4-bromo-4-methyl-1-pentene
CH
3
Br
−
CH
3
CCH
2
CH CH
2
CH
3
Br
106.
His recrystallization was not successful. Because maleic anhydride is a dienophile, it reacts with cyclopen-
tadiene in a Diels–Alder reaction.
+
+
107.
We saw in Problem 106 that maleic anhydride reacts with cyclopentadiene. The function of maleic anhy-
dride in this reaction is to remove cyclopentadiene because removal of a product drives the equilibrium
toward products. (See Le Châtelier’s principle on page 205 of the text.)
108.
The bridgehead carbon cannot have the 120
°
bond angle required for the
sp
2
carbon of a double bond
because, if it did, the compound would be too strained to exist.
















