
306
Copyright © 2017 Pearson Education, Inc.
I. Using Molecular Orbital Theory to Describe Covalent Bond Formation
A. Each of the Overlapping Atomic Orbitals Contributes One Electron to the Bond
Take a look at Figure 1.3 on page 23 of the text. There you can see that an H—H bond is formed by the overlap of
a 1
s
atomic orbital of a hydrogen atom with a 1
s
atomic orbital of another hydrogen atom; each of the atomic orbitals
contributes one electron to the molecular orbital.
SPECIAL TOPIC II
Molecular Orbital Theory
Chemists use models to describe such things as the bonding in molecules, the stability of molecules, and the
reactions between molecules. The model used is generally the one that provides the best description of the molecule
under consideration. One very powerful model is molecular orbital theory.
You were introduced to molecular orbital (MO) theory in Section 1.6 where you saw that electrons are assigned
to a volume of space called an orbital. According to MO theory, covalent bonds are formed when atomic orbitals
combine to form molecular orbitals. Let’s review some important principles:
1.
Orbitals are conserved. In other words, the number of molecular orbitals formed must equal the number
of atomic orbitals combined. For example, when two atomic orbitals interact, two molecular orbitals
are formed—one lower in energy (a bonding MO) and one higher in energy (an antibonding MO,
indicated by a *) than the interacting orbitals.
2.
Electrons always occupy the available
atomic orbitals
with the lowest energy, and no more than two
electrons can occupy an
atomic orbital
. Similarly, electrons always occupy the available
molecular
orbitals
with the lowest energy, and no more than two electrons can occupy a
molecular orbital
.
3.
The relative energies of the molecular orbitals are
s
6
p
6
p
*
6
s
*
.
4.
The strongest covalent bonds are formed by electrons that occupy the molecular orbitals with the lowest
energy. For example, the energy of a
s
MO is lower than that of a
p
MO, and we have seen that a
s
bond is stronger than a
p
bond (Section 1.15).
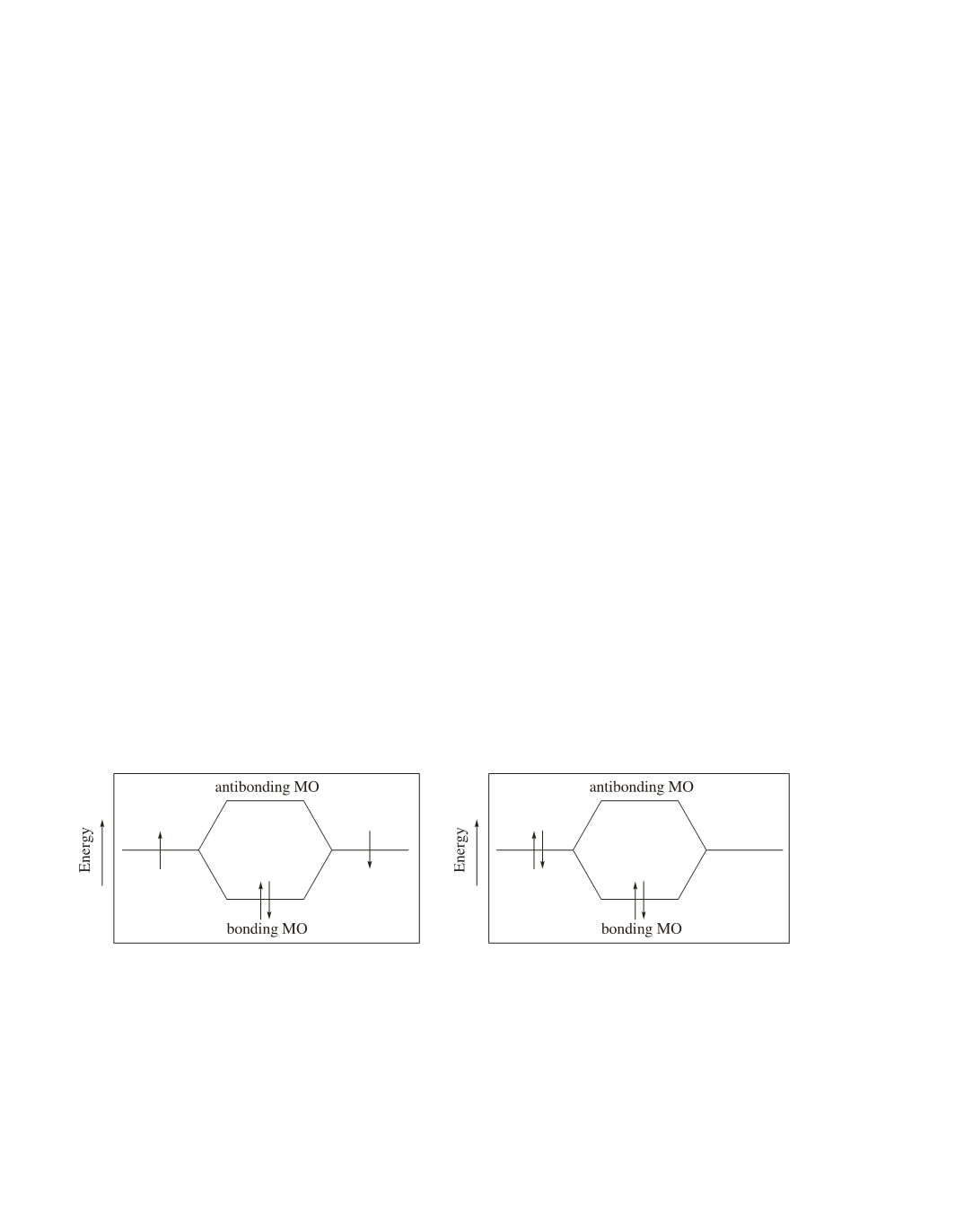
Atomic orbitals can overlap to form molecular orbitals in two ways; these are shown in Figures 1 and 2.
In Figure 1, each of the overlapping atomic orbitals contributes one electron to the bond.
In Figure 2, a filled atomic orbital (it has two electrons) overlaps an empty atomic orbital.
In each case, we see that the electrons are stabilized (are lower in energy) as a result of orbital overlap.
Figure 2. A bond is formed as a result of the overlap
of a filled atomic orbital with an empty atomic orbital.
Figure 1. Each of the overlapping atomic orbitals
contributes one electron to the bond.
















