
Special Topic II
307
Copyright © 2017 Pearson Education, Inc.
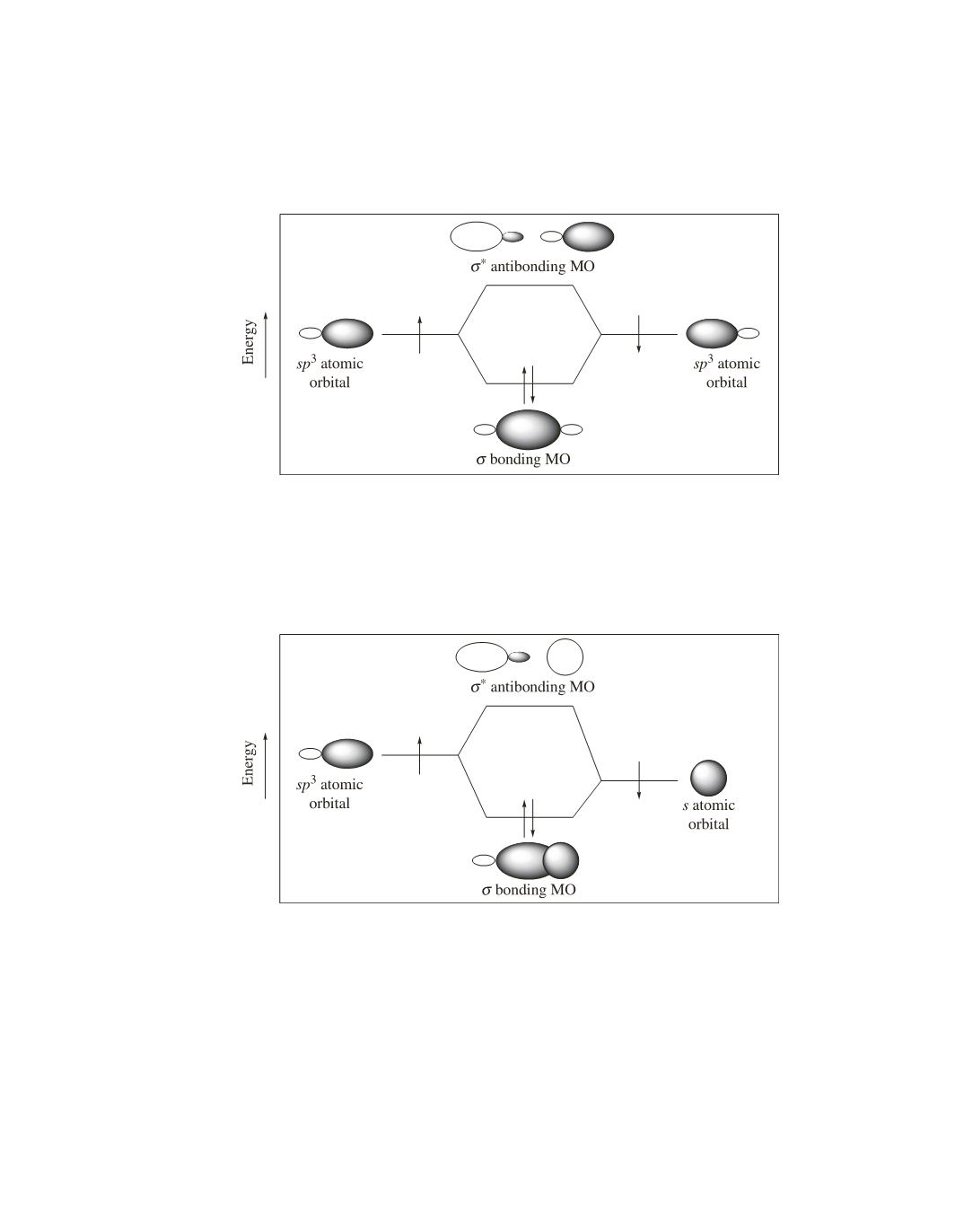
Figure 3 shows that the C—C bond in ethane is formed by the overlap of an
sp
3
atomic orbital of carbon with
an
sp
3
atomic orbital of another carbon; again, each of the overlapping atomic orbitals contributes one electron to
the bond.
Figure 3. The C—C bond of ethane is formed by
sp
3
—
sp
3
overlap.
Figure 4. A C—H bond of ethane is formed by
sp
3
—s overlap.
Figure 4 shows that the C—H bond of ethane is formed by the overlap of an
sp
3
atomic orbital of carbon with
an
s
atomic orbital of hydrogen. Because an
s
atomic orbital of hydrogen is more stable than an
sp
3
atomic orbital
of carbon (see page 27 of the text), the MO formed by
sp
3
—
s
overlap is more stable than the MO formed by
sp
3
—
sp
3
overlap. As a result, the C—H bond is stronger (and shorter) than the C—C bond.
Figure 5 shows that the two
sp
3
atomic orbitals that overlap to form the C—O bond of an alcohol or of an ether
do not have the same energy. An electron is more stable in the atomic orbital of the more electronegative atom.
Thus, the C—O bond is a little stronger and shorter than the C—C bond.
















