
308 Special Topic II
Copyright © 2017 Pearson Education, Inc.
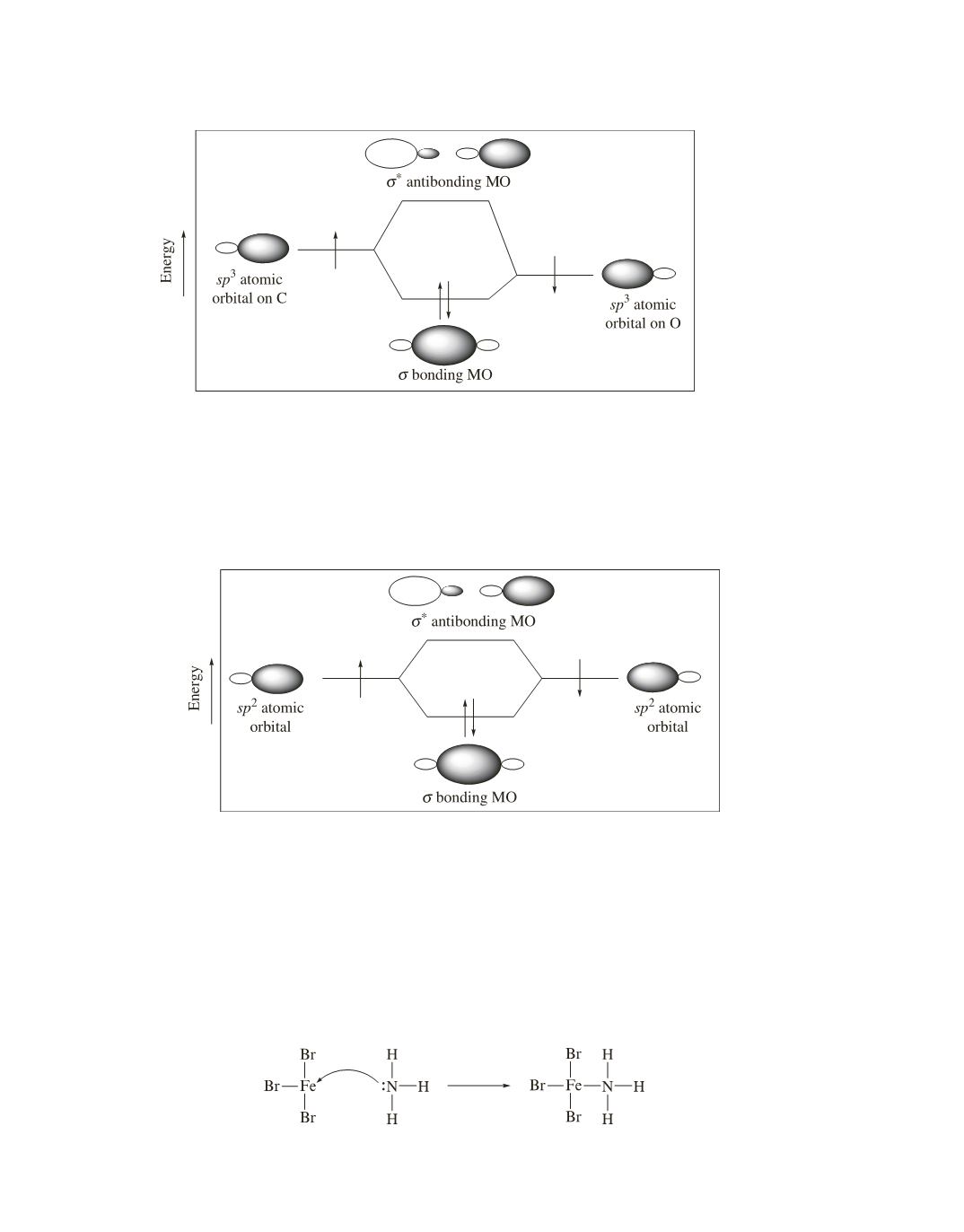
Figure 6 shows that the C—C
s
bond of ethene is formed by the overlap of an
sp
2
atomic orbital of carbon
with an
sp
2
atomic orbital of another carbon. The
p
bond of ethene is formed by the side-to-side overlap of two
p
orbitals. (See Figure 1.4 on page 25 of the text.) A
p
molecular orbital is less stable than a
s
molecular orbital.
The
p
bond, therefore, is weaker than the
s
bond.
Figure 5. The C—O bond of an ether or an alcohol is formed by
sp
3
—
sp
3
overlap.
Figure 6. The C—C
s
bond of ethene is formed by
sp
2
—
sp
2
overlap.
B. A Filled Atomic Orbital Overlaps an Empty Atomic Orbital to Form a Bond
The overlap of a filled atomic orbital with an empty atomic orbital is the second way two atomic orbitals can
overlap to form a bond (Figure 2).
For example, the bond formed between a Lewis base (such as NH
3
) and a Lewis acid (such as FeBr
3
) results
from the base sharing a pair of electrons with the acid. Bond formation results from the overlap of a filled
sp
3
orbital of nitrogen with an empty orbital of iron. This type of reaction is discussed in Section 2.12 of the text.
+
+ −
















