
Special Topic II
309
Copyright © 2017 Pearson Education, Inc.
II. Using Molecular Orbital Theory to Describe Chemical Reactions
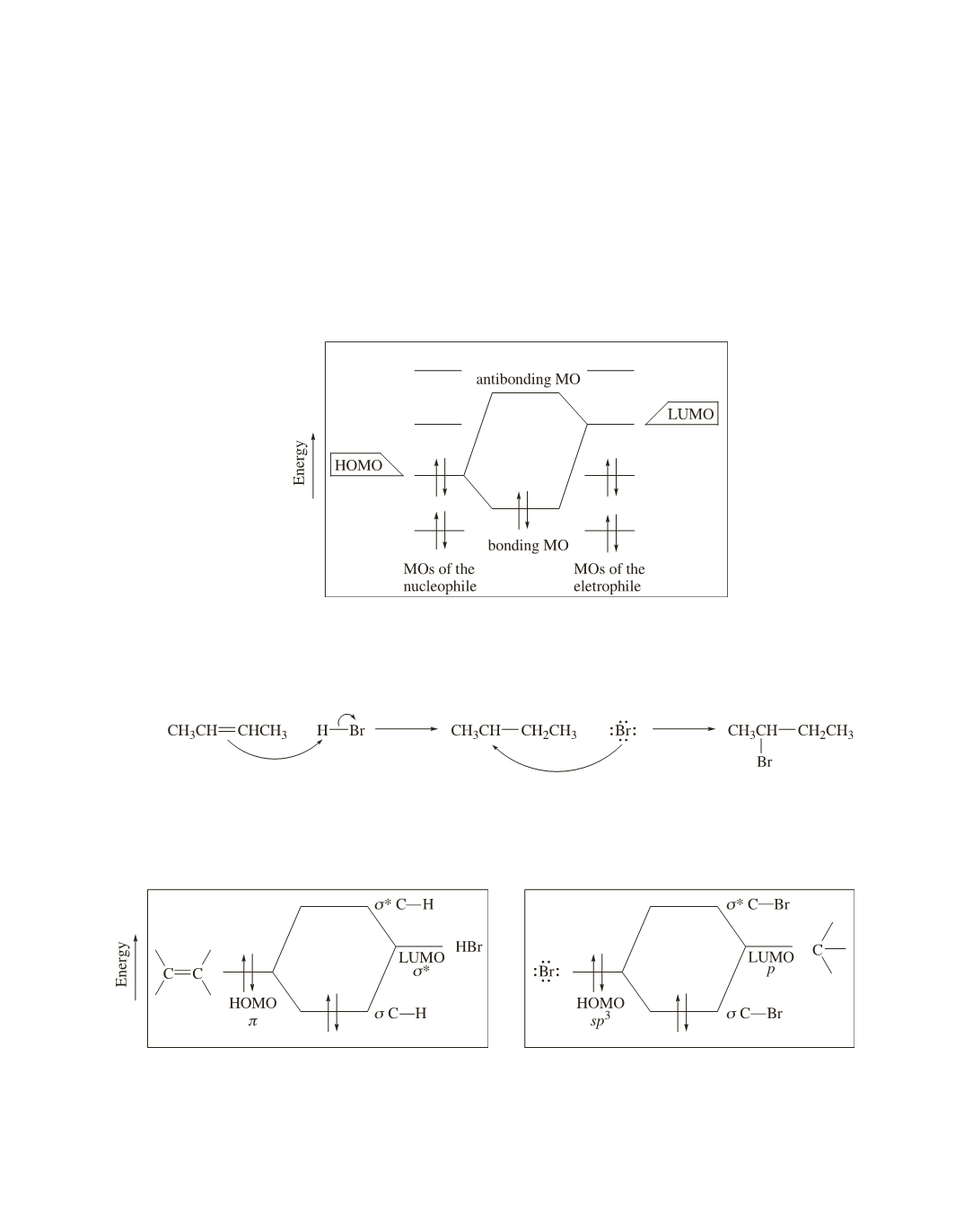
We have seen that most organic reactions involve the reaction of a nucleophile with an electrophile. Molecular
orbital theory describes a reaction between a nucleophile and an electrophile as the result of the interaction of the
HOMO (highest occupied MO) of the nucleophile with the LUMO (lowest unoccupied MO) of the electrophile,
because the most stabilizing interaction is between orbitals closest in energy. Notice that in these examples, a filled
orbital overlaps an empty orbital. The interaction of a filled orbital with an empty orbital is stabilizing, because
the two electrons involved in bond formation end up in the lower-energy bonding MO and no electrons have to be
placed in an antibonding MO (Figure 7).
Figure 7. The interaction of the HOMO of the nucleophile with the LUMO of the electrophile.
We will start by looking at the reaction of 2-butene with HBr, an electrophilic addition reaction we first
examined in Section 5.5.
+
+
+
−
In the first step of the reaction, the alkene is the nucleophile; the electrons of the
p
bond are in the
p
bonding
MO; this is the HOMO. HBr is the electrophile. The electrons that form the H—Br bond are in a
s
bonding MO.
Therefore, the LUMO of HBr is the
s
*
antibonding MO (Figure 8).
Figure 8. Interaction of the HOMO of the alkene with
the LUMO of HBr.
−
+
Figure 9. Interaction of the HOMO of Br
-
with the
LUMO of the carbocation.
















