
310 Special Topic II
Copyright © 2017 Pearson Education, Inc.
In the second step of the reaction, Br
-
is the nucleophile; its HOMO is a filled nonbonding
sp
3
orbital. The
carbocation is the electrophile; the LUMO is carbon’s empty
p
orbital (Figure 9).
In Section 9.1 of the text, we use MO theory to explain why the alkyl halide in an S
N
2 reaction undergoes
back-side attack, resulting in inversion of configuration. The filled nonbonding orbital of the nucleophile is the
HOMO. The LUMO of the electrophilic alkyl halide is the
s
*
antibonding MO, which has its largest lobe at the
back of the carbon in the C—Br bond, so this is where overlap is best. (See Figure 9.1 on page 395 of the text.)
III. Molecular Orbital Theory and Delocalized Electrons
Molecular orbital theory is useful to describe compounds with delocalized electrons. For example, in Section 6.2,
we saw that tertiary carbocations are more stable than secondary carbocations, which are more stable than primary
carbocations, because the electrons in a filled
s
bond are delocalized by overlapping the empty
p
orbital of the
positively charged carbon. (See Figure 6.1 on page 238 of the text.) This kind of electron delocalization is known
as hyperconjugation; it is one more example of the stabilization that results when a filled orbital overlaps an
empty orbital.
Molecular orbital theory avoids having to use contributing resonance structures, because the electrons in the
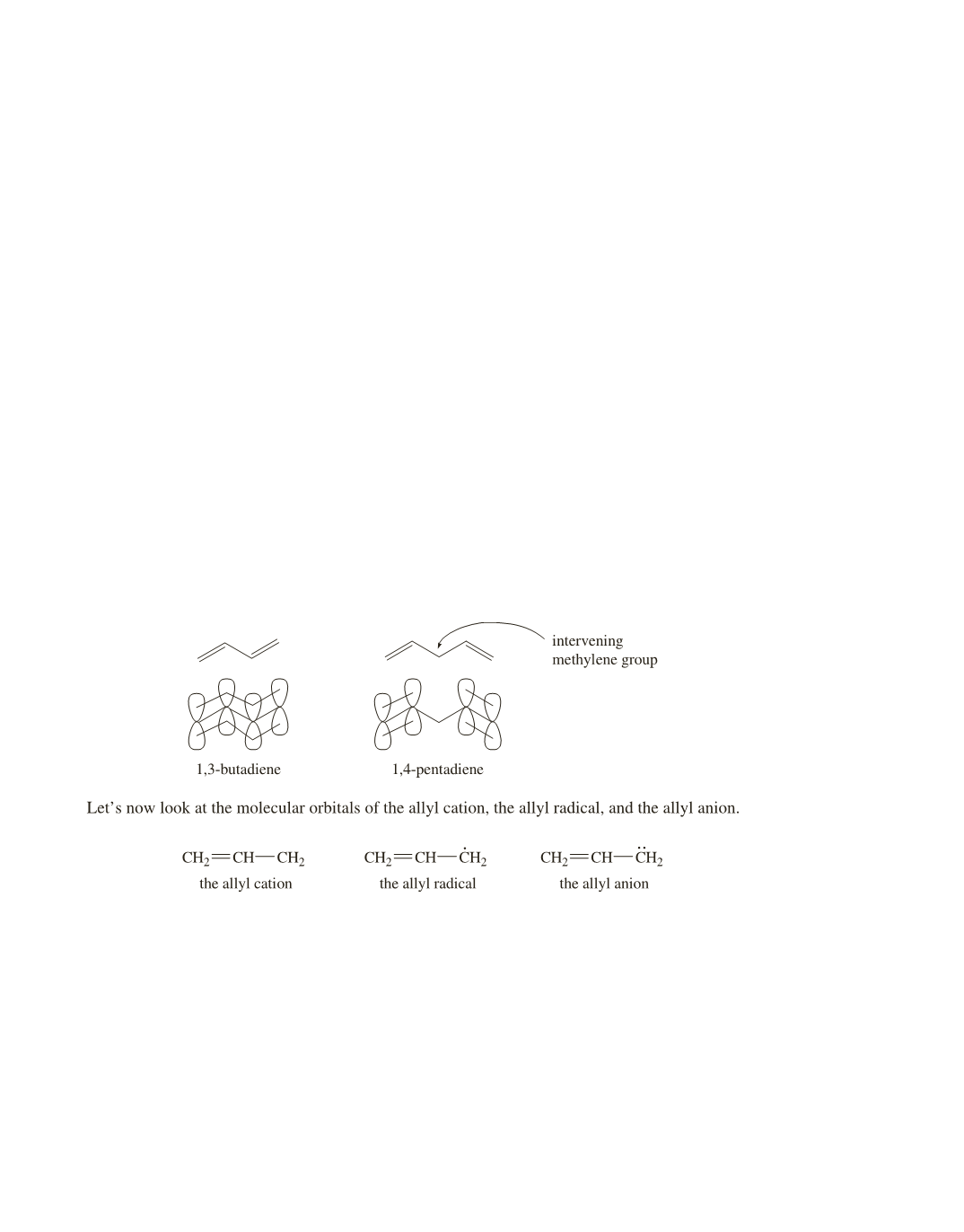
most stable MO are delocalized over the entire molecule. For example, in Section 8.8, we saw that 1,3-butadiene
is more stable than 1,4-pentadiene, because the
p
electrons in 1,3-butadiene are delocalized over four
sp
2
carbons,
whereas the intervening methylene group in 1,4-pentadiene prevents the
p
orbitals of C-2 and C-4 from overlapping.
Thus, the
p
electrons of 1,4-pentadiene are localized, causing its molecular orbitals to have the same energy as
those of ethene—another compound with localized electrons. (See page 337 of the text.)
+
−
















