
Chapter 8 295
Copyright © 2017 Pearson Education, Inc.
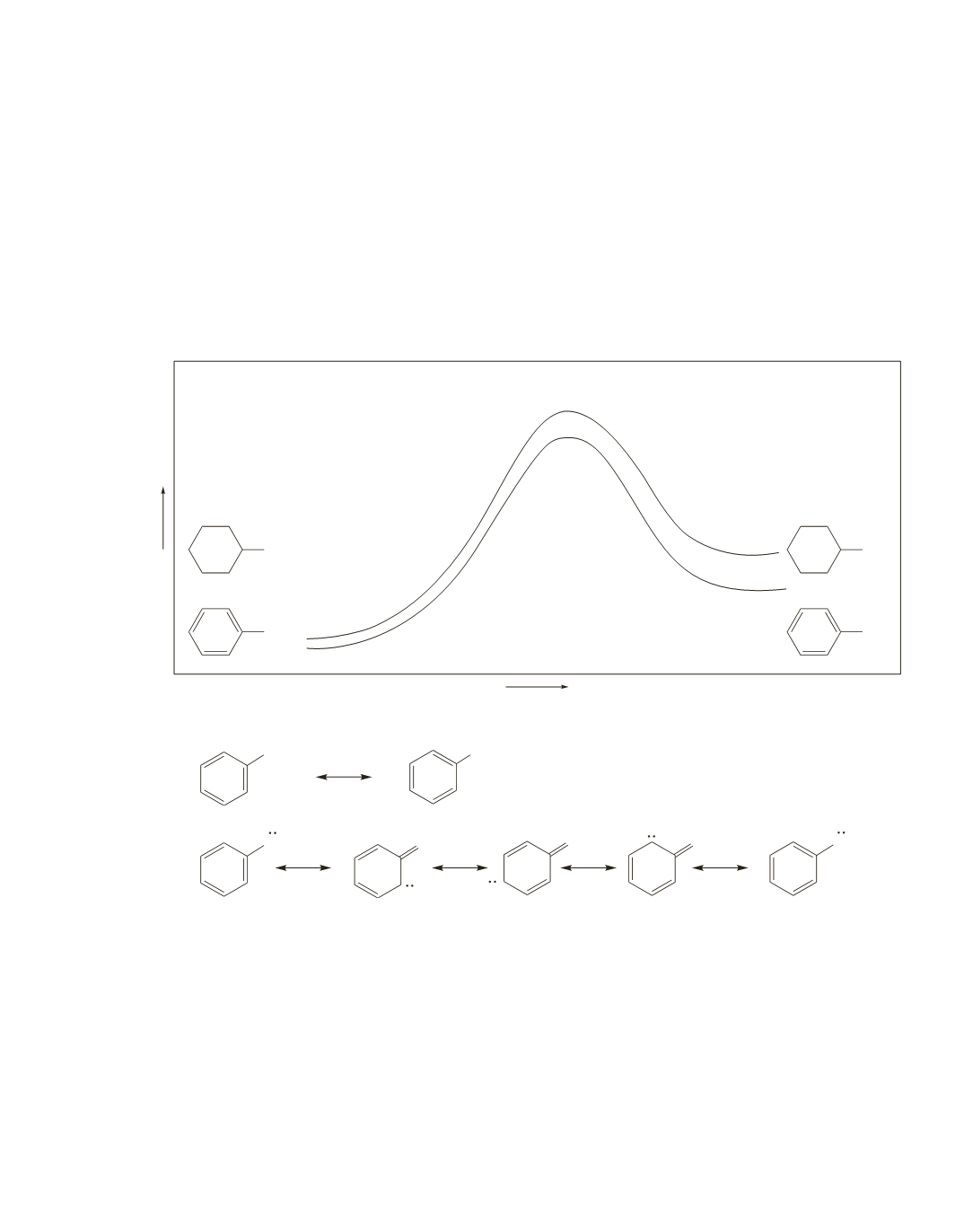
d.
The 2nd, 3rd, and 4th resonance contributors in
c
are more stable than those resonance contributors
in
b
, because in
b
, a positive charge is on the most electronegative atom (the oxygen) and there is
charge separation. Therefore, the phenolate ion has greater electron delocalization than phenol.
Thus, as shown in the energy diagram, the difference in energy between the phenolate ion and the
cyclohexoxide ion is greater than the difference in energy between phenol and cyclohexanol.
e.
Because of greater electron delocalization in the phenolate ion compared to that in phenol, phenol has
a larger
K
a
than cyclohexanol.
f.
Because it has a larger
K
a
(a lower p
K
a
), phenol is a stronger acid.
95.
NH
3
+
NH
3
+
NH
2
NH
2
Free energy
Progress of the reaction
b.
NH
3
NH
3
+
+
c.
NH
2
NH
2
NH
2
NH
2
NH
2
_
_
_
+
+
+
d.
Aniline has greater electron delocalization than the anilinium ion. Therefore, in the energy diagram,
the difference in energy between aniline and cyclohexylamine is greater than the difference in energy
between the anilinium ion and the cyclohexylammonium ion.
e.
Because of greater electron delocalization in aniline compared to that in the anilinium ion, the ani-
linium ion has a larger
K
a
than the cyclohexylammonium ion.
f.
Because it has a larger
K
a
(a lower p
K
a
), the anilinium ion is a stronger acid than the cyclohexylam-
monium ion. Therefore, cyclohexylamine is a stronger base than aniline. (The stronger the acid, the
weaker its conjugate base.)
















