
290 Chapter 8
Copyright © 2017 Pearson Education, Inc.
83.
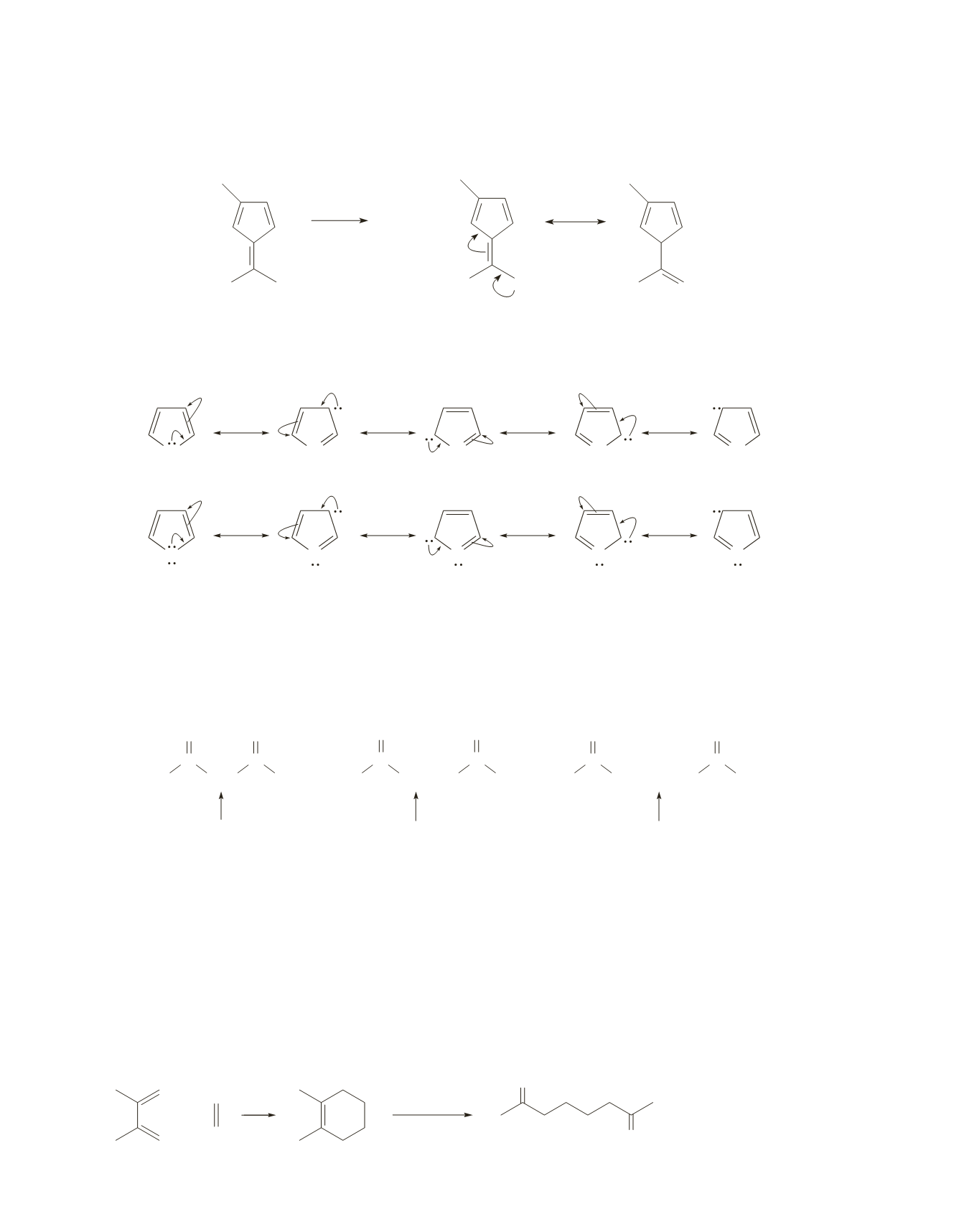
The following compound is the strongest acid, because it is the only one that forms a conjugate base that is
aromatic. Recall that the more stable (weaker) the base, the stronger is its conjugate acid.
..
_
..
_
aromatic
H
+
+
84.
The resonance contributors of pyrrole are more stable because the positive charge is on nitrogen. In furan,
the positive charge is on oxygen, which, being more electronegative, is less stable with a positive charge.
N
H
N
H
N
H
N
H
N
H
O
O
O
O
O
+
+
+
+
+
+
+
+
−
−
−
−
−
−
−
−
85.
A
is the most acidic because the electrons left behind when the proton is removed can be delocalized onto
two oxygens.
B
is the next most acidic because the electrons left behind when the proton is removed can be delocalized
onto one oxygen.
C
is the least acidic because the electrons left behind when the proton is removed cannot be delocalized.
CH
3
C
O
CH
2
C
CH
3
O
CH
3
C
O
CH
2
CH
2
C
CH
3
O
CH
3
C
O
CH
2
CH
2
CH
2
C
CH
3
O
A
B
C
>
>
86.
a.
It has eight molecular orbitals.
b.
c
1
,
c
2
,
c
3
, and
c
4
are bonding molecular orbitals;
c
5
,
c
6
,
c
7
, and
c
8
are antibonding molecular
orbitals.
c.
c
1
,
c
3
,
c
5
, and
c
7
are symmetric molecular orbitals;
c
2
,
c
4
,
c
6
, and
c
8
are antisymmetric molecular
orbitals.
d.
c
4
is the HOMO and
c
5
is the LUMO in the ground state.
e.
c
5
is the HOMO and
c
6
is the LUMO in the excited state.
f.
If the HOMO is symmetric, the LUMO is antisymmetric and vice vers
a.
g.
It has seven nodes between the nuclei. It also has one node that passes through the nuclei.
87.
+
O
O
1. O
3
, −78
°
C
2.
(
CH
3
)
2
S
















