
288 Chapter 8
Copyright © 2017 Pearson Education, Inc.
77.
a.
+
O
O
O
+
O
O
O
CH
3
CH
2
O
CH
3
O
CH
3
O
CH
3
CH
2
O
O
O
b.
78.
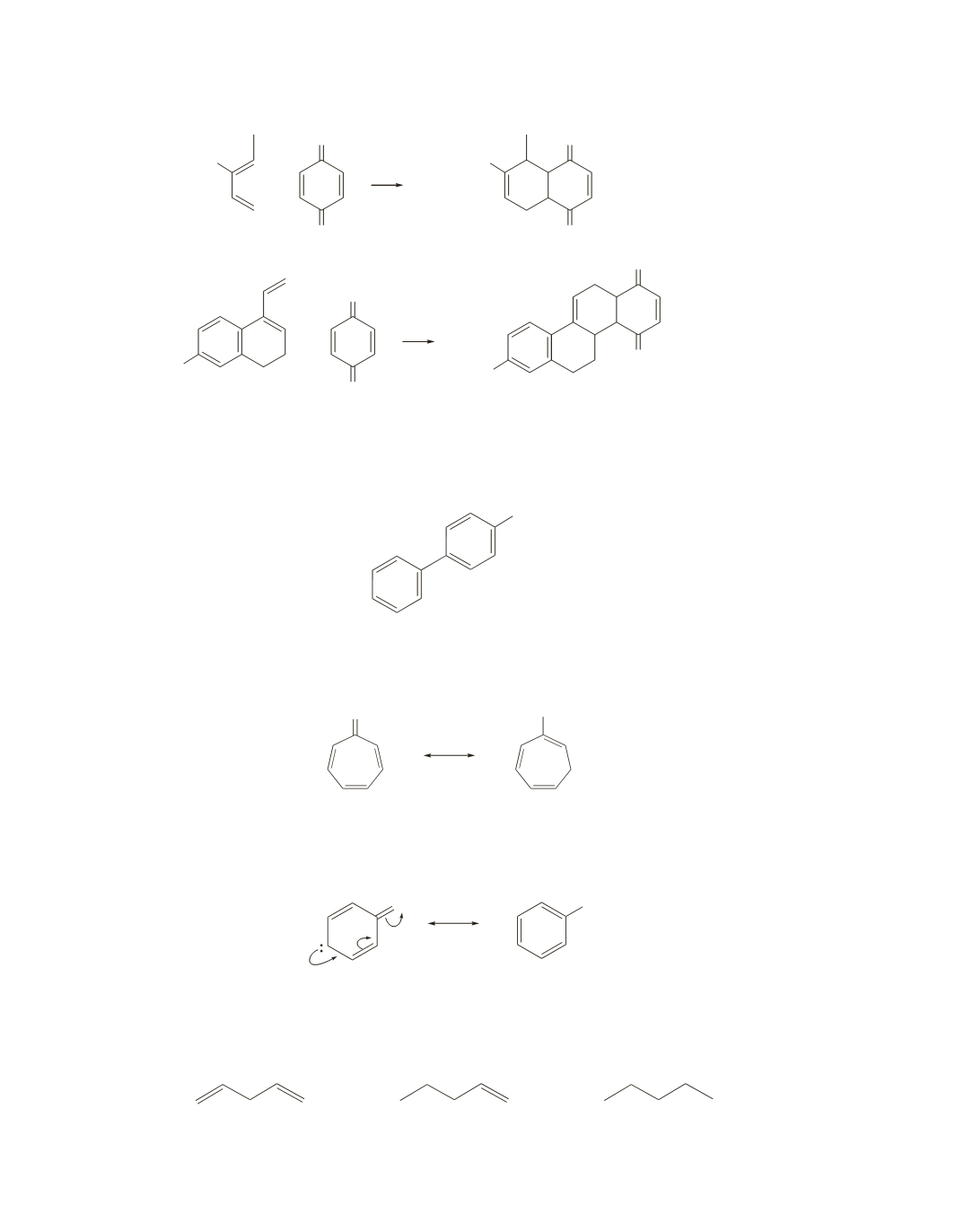
a.
The structure shown below is the stronger acid because it has the weaker conjugate base. When this
compound loses a proton, the electrons left behind can be delocalized onto six different carbons. The
electrons left behind on the conjugate base of the other compound can be delocalized onto only three
different carbons; they cannot be delocalized into the second benzene ring.
OH
b. 1.
The first compound has the greater electron density on its oxygen because the resonance contribu-
tor with a negative charge on oxygen is particulalry stable since it is aromatic.
O
O
_
+
aromatic
2.
The first compound has the greater electron density on its oxygen because the resonance contribu-
tor with a negative charge on oxygen is particularly stable since it is is aromatic.
O
−
O
−
aromatic
79.
The first carbocation is the most stable because its positive charge can be shared by two other carbons; the
positive charge on the next carbocation can be shared by one other carbon; the positive charge on the base
on the right cannot be shared by other carbons.
>
>
+
+
+
















