
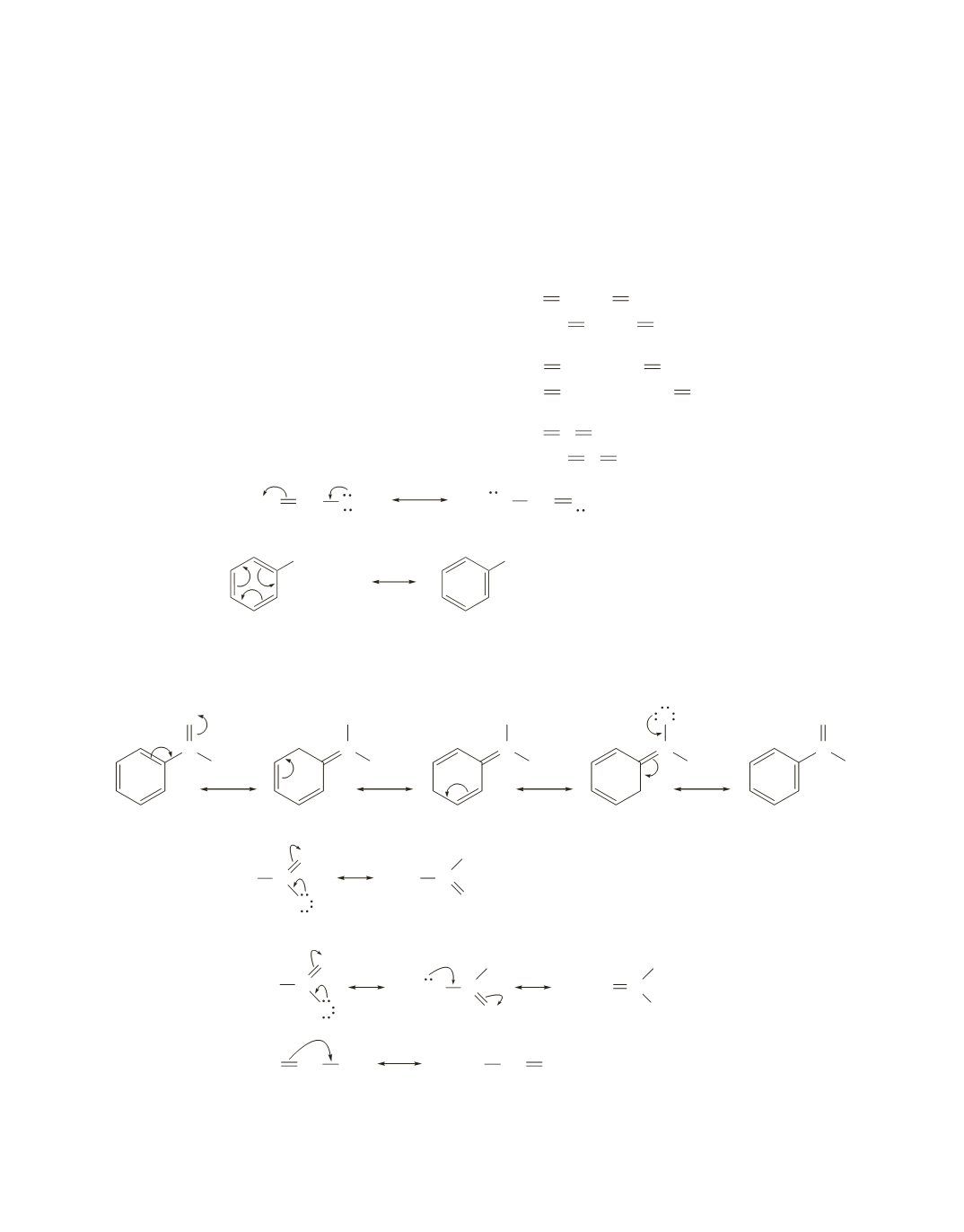
Chapter 8 283
Copyright © 2017 Pearson Education, Inc.
66.
a.
different compounds
b.
different compounds
c.
resonance contributors
d.
resonance contributors
e.
different compounds
Notice that in the structures that are different compounds, both atoms and electrons have changed their
locations. In the structures that are resonance contributors, only the electrons have moved.
67.
a.
There are six linear dienes with molecular formula C
6
H
10
.
b.
Two are conjugated dienes.
c.
Two are isolated dienes.
d.
Two are cumulated dienes.
CH
2
CHCH CHCH
2
CH
3
CH
3
CH CHCH CHCH
3
CH
2
CHCH
2
CH CHCH
3
CH
2
CHCH
2
CH
2
CH CH
2
CH
2
C
CHCH
2
CH
2
CH
3
CH
3
CH C CHCH
2
CH
3
68.
a. 1.
CH
3
CH CH OCH
3
CH
3
CH CH OCH
3
−
+
minor
major
2.
CH
2
NH
2
CH
2
NH
2
The two resonance contributors have the same stability and, therefore, contribute equally to the
resonance hybrid.
3.
CH
3
C
O
CH
3
C
O
−
CH
3
C
O
−
CH
3
C
O
CH
3
C
O
+
+
+
−
minor
minor
minor
major
major
4.
CH
3
CH
3
The two resonance contributors have the same stability and, therefore, contribute equally to the hybrid.
N
O
O
−
+
N
O
−
O
+
5.
CH
3
CH N
O
minor
−
+
CH
3
CH N
O
−
O
minor
−
+
CH
3
CH N
O
−
O
−
major
+
O
−
6.
+
3
CH CH CH CH
minor
major
2
+
3
CH CH CH CH
2
















