
Chapter 8 275
Copyright © 2017 Pearson Education, Inc.
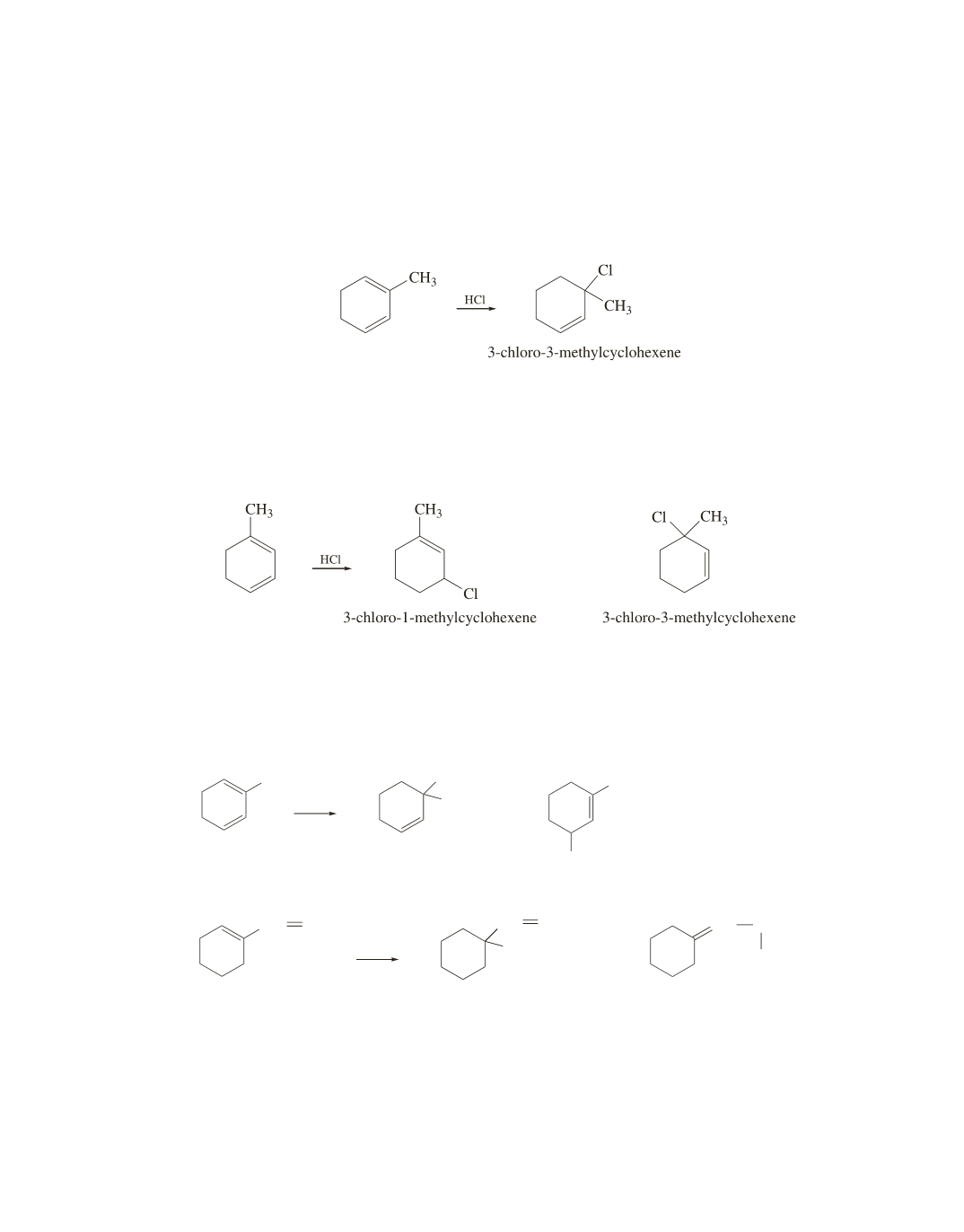
34.
She should follow her friend’s advice. If she uses 2-methyl-1,3-cyclohexadiene, the fastest formed prod-
uct will be 3-chloro-3-methylcyclohexene, both if the proximity effect controls which product is formed
faster
and
if the more stable transition state controls which product is formed faster, because this product
is formed through a transition state in which the positive charge is primarily on a tertiary allylic carbon.
Therefore, the experiment will not be able to establish which of the two effects controls which product is
formed faster.
If she follows her friend’s advice and uses 1-methyl-1,3-cyclohexadiene, the faster-formed product will be
3-chloro-1-methylcyclohexene only if the proximity effect controls which product is formed faster. The
product will be 3-chloro-3-methylcyclohexene if the more stable transition state controls which product is
formed faster, because only this product is formed through a transition state in which the positive charge is
primarily on a tertiary allylic carbon.
+
35.
a.
The rate-determining step is formation of the carbocation.
b.
The product-determining step is reaction of the carbocation with the nucleophile.
36.
Solved in the text.
37.
a.
CH
3
HCl
CH
3
Cl
+
Cl
CH
3
kinetic product
thermodynamic product
CH
HCl
Cl
CH
+
CH
kinetic product
thermodynamic product
CHCH
3
CHCH
3
CHCH
3
Cl
b.
Notice that the 1,2-product is always the kinetic product.
The thermodynamic product is the product with the most highly substituted double bond.
















