
Chapter 8 273
Copyright © 2017 Pearson Education, Inc.
CH
3
CHCHCH CHCH
3
Cl Cl
CH
3
CHCH CHCHCH
3
1,2-addition product
1,4-addition product
Cl
CH
3
CH
2
C C
Br
CH
3
CH
3
CHCH
3
1,2-addition product
Br
Br
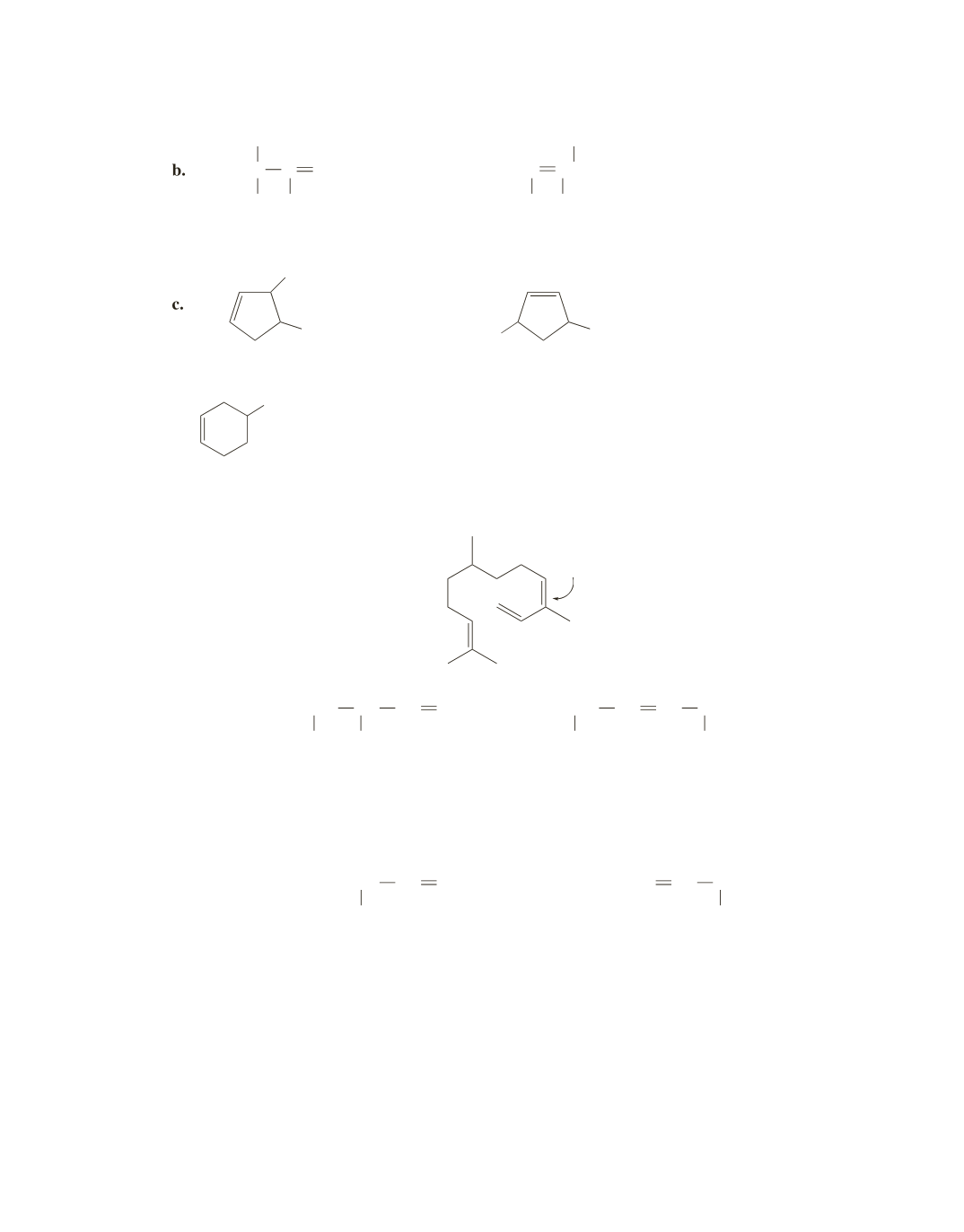
+
+
+
1,2-addition product
Br
Br
1,4-addition product
Cl
CH
3
CH
2
C CCHCH
3
Br
CH
3
CH
3
1,4-addition product
d.
Cl
29.
The indicated double bond is the most reactive in an electrophilic addition reaction with HBr because addi-
tion of an electrophile to this double bond forms the most stable carbocation (a tertiary allylic cation).
30.
second reaction:
This compound has an asymmetric center,
so both the
R
and
S
stereoisomers
will be obtained. (Note that
E
and
Z
stereoisomers are not possbile for
the double bond.)
This compound has a double bond,
so both the
E
and
Z
stereoisomers
will be obtained.
CH
3
CH CH
*
CH
2
Br
CH
3
CH CH CH
2
Br
This compound has an asymmetric center,
so both the
R
and
S
stereoisomers
will be obtained. (Note that
E
and
Z
stereoisomers are not possbile for
the double bond.)
rst reaction:
This compound has a double bond,
so both the
E
and
Z
stereoisomers
will be obtained.
CH
2
CH CH
*
CH
2
Cl
Cl
CH
2
CH CH CH
2
Cl
Cl
















