
740 Chapter 22
Copyright © 2017 Pearson Education, Inc.
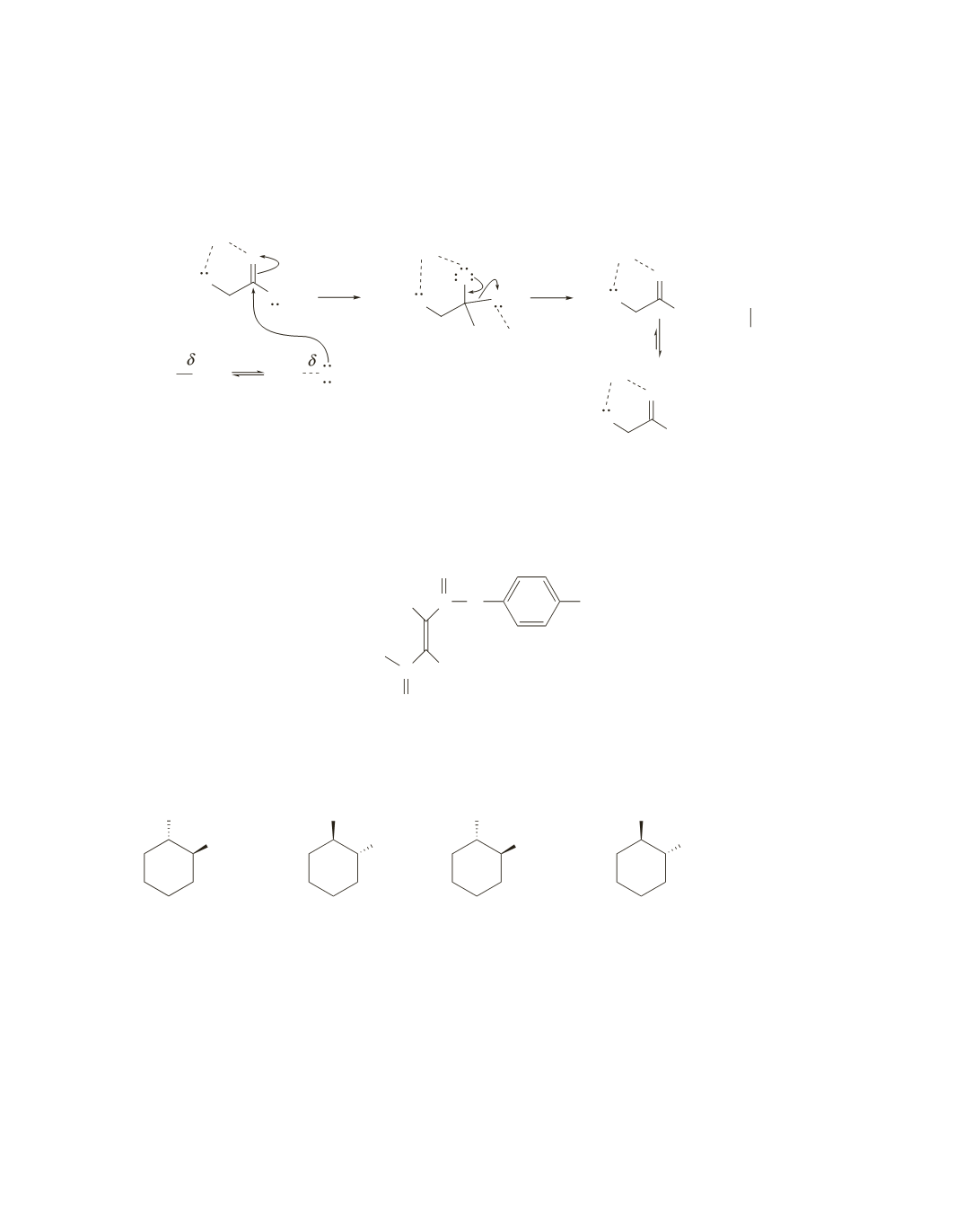
8.
Co
2
+
can catalyze the reaction in three different ways. It can complex with the reactant, increasing the
susceptibility of the carbonyl group to nucleophilic addition. It can also complex with water, increasing the
tendency of water to lose a proton, resulting in a better nucleophile for hydrolysis. And it can complex with
the leaving group, decreasing its basicity and thereby making it a better leaving group.
+
NH
3
+
Co
2+
Co
+
Co
+
OH
2
Co
+
OH
+
H
+
+
−
+
NH
2
H
2
N
NH
2
O
Co
+
H
2
N
OH
O
H
2
N
NH
2
O
Co
+
Co
+
Co
+
Co
+
HO
H
2
N
O
O
_
_
9.
Because the reacting groups in the trans isomer are pointed in opposite directions, they cannot react in an
intramolecular reaction. Because they can react only via an intermolecular pathway, they will have approx-
imately the same rate of reaction as they would have if the reacting groups were in separate molecules.
Consequently, the relative rate would be expected to be close to one.
Br
OC
O
H
C H
O
−
O
10.
a.
The nucleophile can attack the back side of either of the two ring carbons to which the sulfur is bonded
in the intermediate, thereby forming two trans products. There are two nucleophiles (water and etha-
nol), so a total of four products will be formed.
b.
OH
SC
6
H
5
SC
6
H
5
OH
OCH
2
CH
3
SC
6
H
5
SC
6
H
5
OCH
2
CH
3
11.
Solved in the text.
12.
The tetrahedral intermediate has two leaving groups, a carboxylate ion and a phenolate ion. When there are
no nitro substituents on the benzene ring, the carboxylate ion is a weaker base (a better leaving group) than
the phenolate ion, so the tetrahedral intermediate reforms the ester. Thus, the reaction proceeds through
general-base catalyzed hydrolysis of the ester.
In contrast, when there are nitro groups on the benzene ring, the 2,4-dinitrophenolate ion is a weaker base
(a better leaving group) than the carboxylate ion, so the tetrahedral intermediate forms the anhydride.
Thus, the reaction proceeds through hydrolysis of the anhydride formed by nucleophilic catalysis.
















