
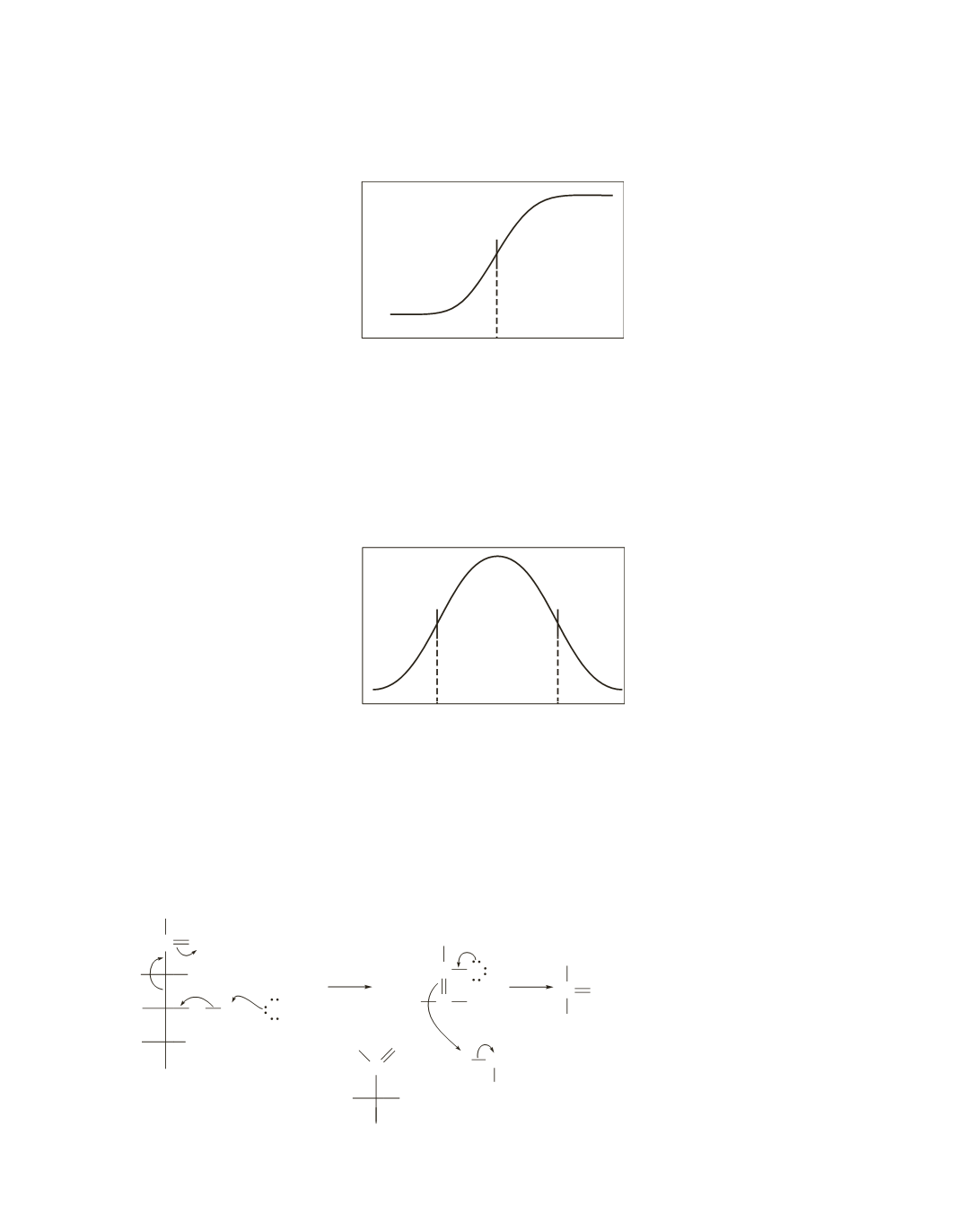
744 Chapter 22
Copyright © 2017 Pearson Education, Inc.
b.
Because the catalytic group is a base catalyst, it will be inactive in its acidic form and active in its basic
form.
enzyme activity
p
K
a
pH
7.2
22.
The ascending leg of the pH-rate profile is due to a group that is a general-base catalyst because the rate is
at a maximum when the group is in its basic form. From the description of the mechanism, we know that
amino acid is histidine.
The descending leg of the pH-rate profile is due to a group that is a general-acid catalyst because the rate is
at a maximum when the group is in its acidic form. From the description of the mechanism, we know that
amino acid is lysine.
enzyme activity
pH
6.7
9.3
p
K
a
p
K
a
23.
In the absence of an enzyme, d-fructose is in equilibrium with d-glucose and d-mannose as a result of
an enediol rearrangement (Section 20.5). Both C-2 epimers are formed, because a new asymmetric cen-
ter is formed at C-2 and it can have either the
R
or the
S
configuration. Enzyme-catalyzed reactions are
typically highly stereoselective—the enzyme catalyzes the formation of a single stereoisomer. Therefore,
d-fructose is in equilibrium with only d-glucose in the presence of the enzyme that catalyzes the enediol
rearrangement.
24.
CH
2
OPO
3
C O
CH
2
OPO
3
H
HO
H O
H OH
HO
H
2
−
2
−
OH
HO C H
C
CH
2
OPO
3
O
2
−
CH
2
OPO
3
H OH
2
−
O
H
−
−
H
C
H O
C
CH
2
OPO
CH
2
OH
3
O
2
−
−
















