
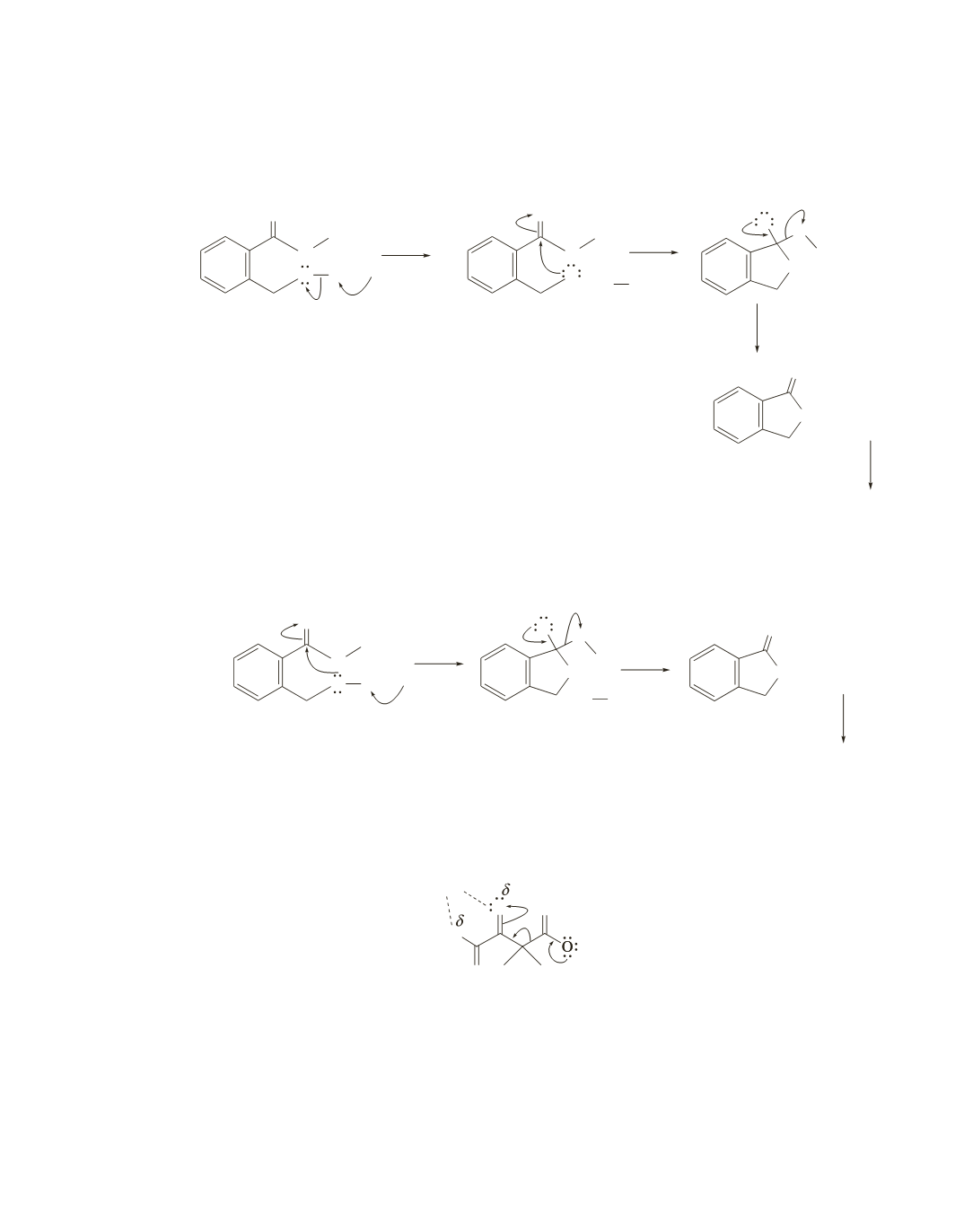
Chapter 22 739
Copyright © 2017 Pearson Education, Inc.
5.
Solved in the text.
6.
a.
In specific-base catalysis, the proton is removed from the reactant before the O
¬
C bond forms.
O
O
O
O
O
O
_
_
+
CH
3
O
_
HB
+
CH
3
OH
+
B
..
H B
+
O
O
O
H B
..
O
O
b.
In general-base catalysis, the proton is removed from the reactant and the O
¬
C bond forms in the
same step.
O H
O
B
..
O
O
_
+
CH
3
O
−
H B
+
HB
+
CH
3
OH
+
B
..
O
O
O
O
7.
The metal ion catalyzes the decarboxylation reaction by complexing with the negatively charged oxygen of
the carboxylate group and carbonyl oxygen of the
b
-keto group, thereby making it easier for the carbonyl
oxygen to accept the electrons that are left behind when CO
2
is eliminated.
O O
O
Cu
+
+
O
−
−
Because acetoacetate and the monoethyl ester of dimethyloxaloacetate do not have a negatively charged
oxygen on one carbon and a carbonyl group on an adjacent carbon with which to form a complex, a metal
ion does not catalyze decarboxylation of these compounds.
















