
Chapter 22 745
Copyright © 2017 Pearson Education, Inc.
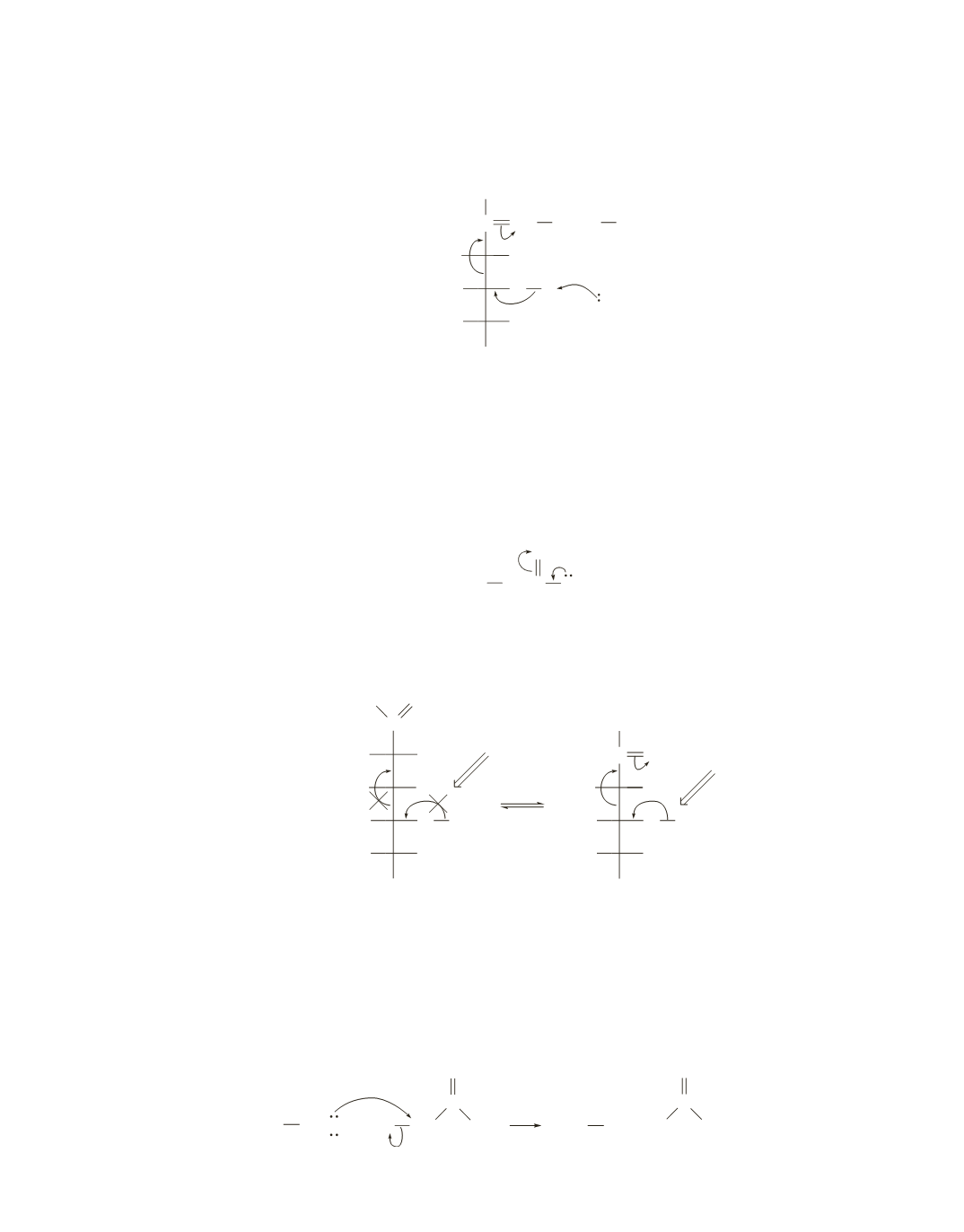
25.
The positively charged nitrogen of the protonated imine can accept the electrons that are left behind when
the C3
¬
C4 bond breaks.
C NH
HO H
H O
H OH
CH
2
OPO
3
H
(CH
2
)
4
Lys
2
−
CH
2
OPO
3
2
−
+
B
In the absence of the protonated imine, the electrons would be delocalized onto a neutral oxygen. (See
Problem 24.) The neutral oxygen is not as electron withdrawing as the positively charged nitrogen of the
protonated imine. In other words, imine formation makes it easier to break the C3
¬
C4 bond.
26.
Because
2
is a primary amine, it can form an imine. Notice that
1
cannot form an imine because the lone
pair on the NH
2
group is delocalized onto the oxygen, so this NH
2
group is not a nucleophile. The N in
3
is
not nucleophilic because its lone pair is delocalized (the lone pair is part of indole’s
p
cloud).
O
CH
2
C NH
2
27.
In order to break the C3
¬
C4 bond, the carbonyl group has to be at the 2-position as it is in fructose so that it
can accept the electrons that result from C3
¬
C4 bond cleavage; the carbonyl group at the 1-position in glucose
cannot accept these electrons. Therefore, glucose must isomerize to a ketose before the reaction can occur.
H OH
HO H
H O
H OH
CH
2
OPO
3
H
2
−
No
CH
2
OH
C O
HO H
H O
H OH
CH
2
OPO
3
H
2
−
glucose-6-phosphate
fructose-6-phosphate
Yes
C
H O
28.
Cysteine residues react with iodoacetic acid because a thiol is a good nucleophile and iodine is a good
leaving group. If a cysteine residue is at the active site of an enzyme, adding a substituent to the sulfur in
this way could interfere with the enzyme’s being able to bind the substrate or it could interfere with posi-
tioning the tyrosine residue that catalyzes the reaction. Adding a substituent to cysteine might also cause a
conformational change in the enzyme that could destroy its activity.
Cys CH
2
SH
+
I
Cys CH
2
S
I
−
+
H
+
+
O CH
2
C
O
O CH
2
C
O
−
−
















