
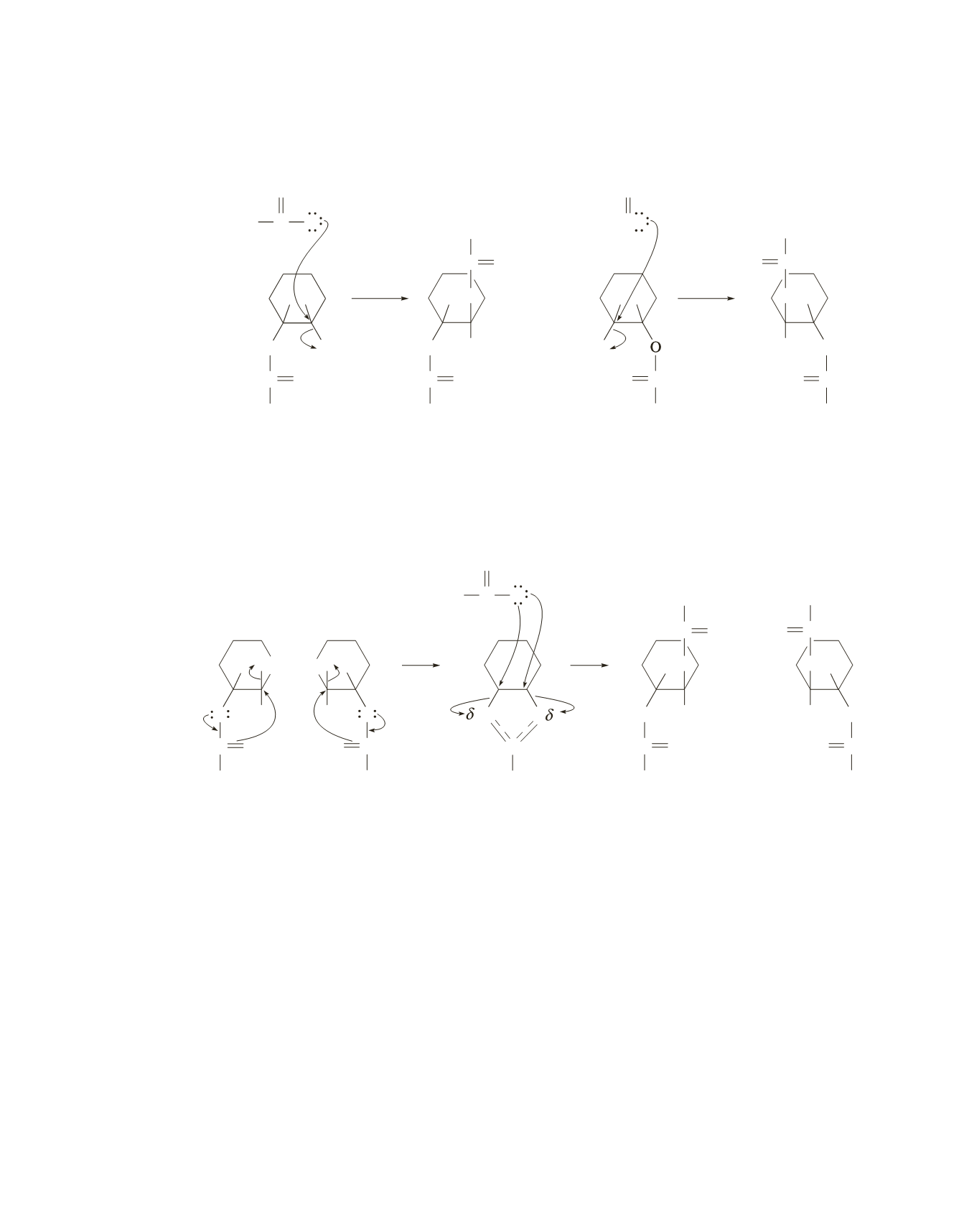
Chapter 22 751
Copyright © 2017 Pearson Education, Inc.
41.
a.
The cis reactants each undergo an S
N
2 reaction. Because the acetate displaces the tosylate group by
back-side attack, each cis reactant forms a trans product.
−
−
O
O
O
O
OTs
C
O
O
O
C
CO
C
O
TsO
O
OC
C
O C
O
O
O
C
CH
3
CH
3
CH
3
CH
3
CH
3
CH
3
CH
3
CH
3
H
H
H
H
H
H
H
H
b.
The acetate group in a trans reactant is positioned to be able to displace the tosylate leaving group by
an intramolecular S
N
2 reaction. Acetate ion then attacks in a second S
N
2 reaction from the back side of
the group it displaces, so trans products are formed. Because both trans reactants form the same inter-
mediate, they both form the same products. Because the acetate ion can attack either of the carbons in
the intermediate equally easily, a racemic mixture will be formed.
+
+
+
−
OTs TsO
O
O
O
a
a b
b
O
O O
O
O
O
O
O
O
O
O
O
O
C
C
C
C
C
C
C
C
CH
3
CH
3
CH
3
CH
3
CH
3
CH
3
CH
3
CH
3
H
H
H
H
H
H
H
H
c.
The trans reactants are more reactive because the tosylate leaving group is displaced in an intra
molecular reaction, and the resulting positively charged cis intermediate is considerably more reactive
than the neutral cis reactants. Thus, a trans isomer undergoes two successive S
N
2 reactions, each of
which is faster than the one step S
N
2 reaction that a cis isomer undergoes.
42.
Reduction of the imine linkage with sodium borohydride causes fructose to become permanently attached
to the enzyme because the hydrolyzable imine bond has been lost. Acid-catalyzed hydrolysis removes the
two phosphate groups and hydrolyzes the two peptide bonds, so the radioactive fragment that is isolated
after hydrolysis is the lysine residue (covalently attached to fructose) that originally formed the imine.
















