
Chapter 22 753
Copyright © 2017 Pearson Education, Inc.
b.
3-Aminoindole would not be as effective a catalyst, because the electrons left behind when CO
2
is
eliminated cannot be delocalized onto an electronegative atom.
+
OH
N C
C O
R
C
C
O
NH
3-aminoindole
-keto acid
imine formation
N
H
N
H
O
H
2
O
R
44.
a.
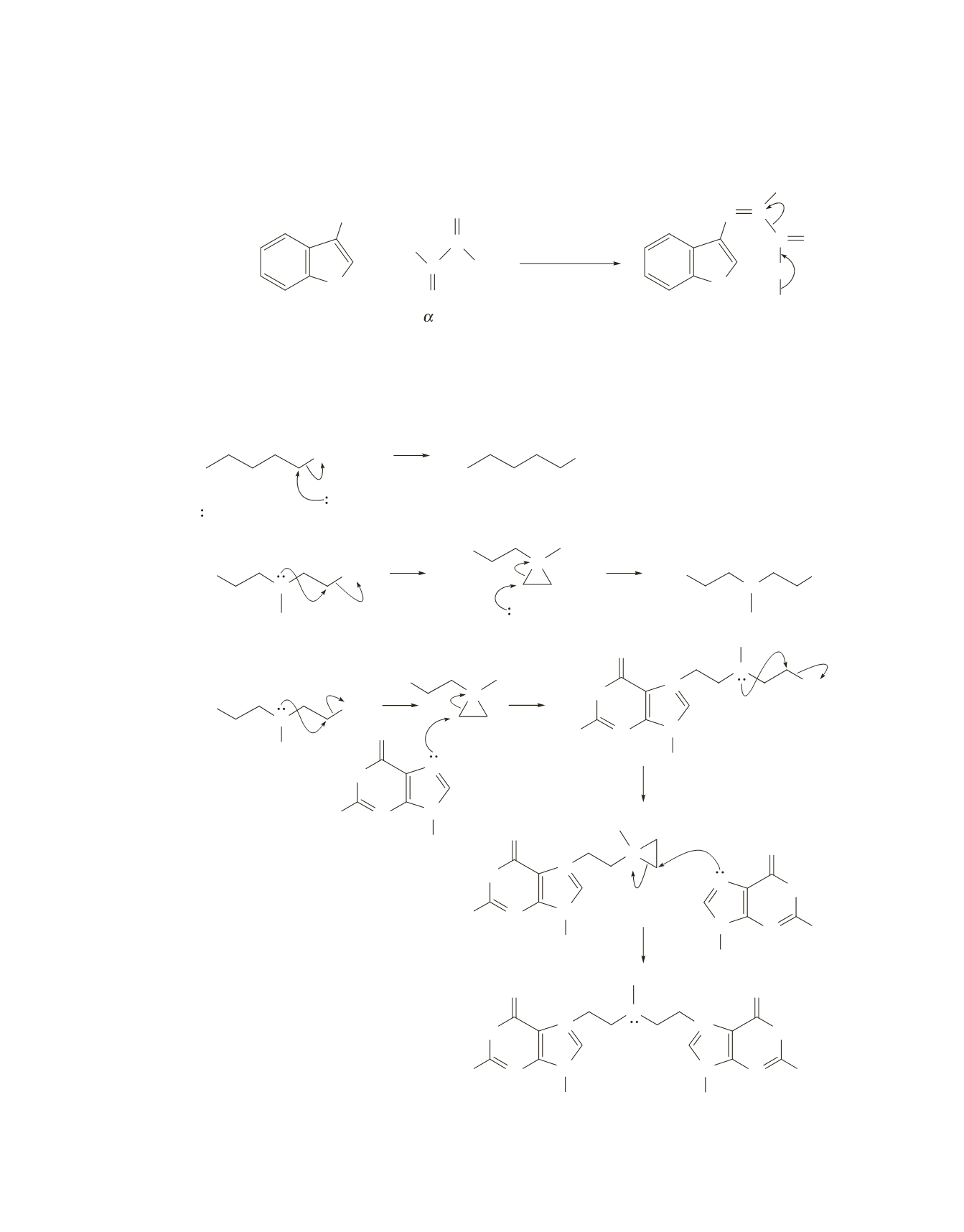
Intramolecular nucleophilic attack on an alkyl halide occurs more rapidly than intermolecular attack on
an alkyl halide, because the reacting groups are tethered in the former, which makes it more likely that
they will find each other to react. The intramolecular reaction is followed by another relatively rapid
S
N
2 reaction, because the strain in the three-membered ring causes it to break easily.
−
Cl
Cl
Nu
=
Nuc
−
Nuc
−
guanine
−
−
+
N
Cl
Cl
Cl
N
Cl
Nuc
N
Nuc
Cl
b.
−
+
N
Cl
Cl
N
Cl
N
N
Cl
+
N
N
+
+
N
N
+
C
l
−
C
l
−
C
l
−
C
l
−
C
l
−
C
l
O
O
O
O
O
R
R
R
R
R
R
O
HN
HN
HN
HN
N
N N
N
N
N
N
N
N
N
NH
NH
NH
2
NH
2
N N
N
N
N
H
2
N
H
2
N
H
2
N
H
2
N
+
















