
Chapter 22 743
Copyright © 2017 Pearson Education, Inc.
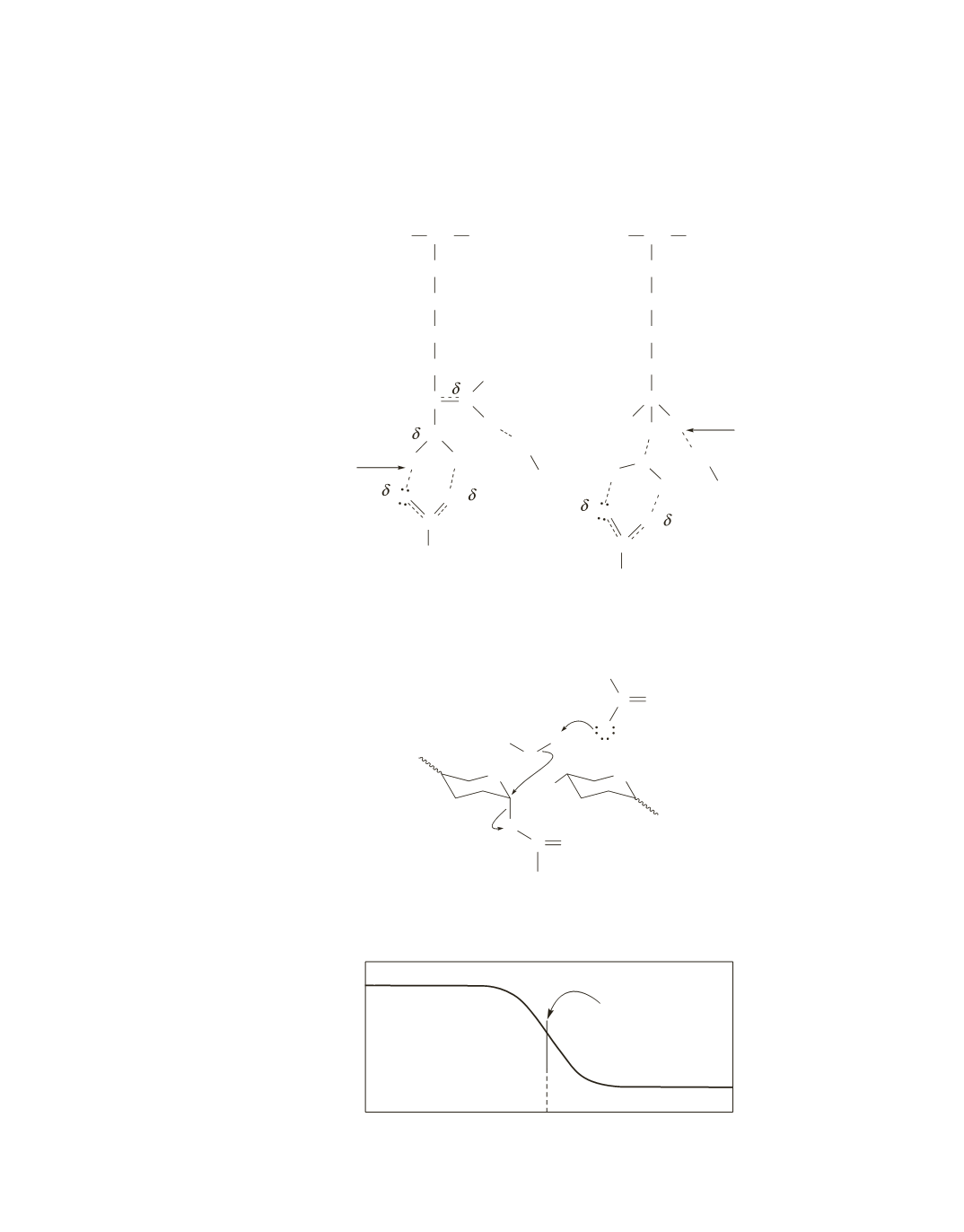
18.
Because arginine extends farther into the binding pocket, it must be the one that forms direct hydrogen
bonds. Lysine, which is shorter, needs the mediation of a water molecule in order to engage in hydrogen
bond formation with aspartate.
CH
CH
2
CH
2
CH
2
NH
C N
N
H
H
H H
−
O O
C
HOCH
2
+
−
+
CH
CH
2
CH
2
CH
2
CH
2
N
HH H end of lysine
end of arginine
O
H
H
O O
C
+
−
−
arginine
lysine
HOCH
2
19.
The side chains of d-Arg and d-Lys are not positioned to bind correctly at the active site.
They would, however, be able to bind at a mirror image of the active site.
20.
NAM will contain
18
O because it is the ring that undergoes nucleophilic attack by H
2
18
O.
−
C O
O
O
HO
O
OC
O
NAM
NAG
O
H H
18
21.
a.
Because the catalytic group is an acid catalyst, it will be active in its acidic form and inactive in its
basic form. The pH at the midpoint of the curve corresponds to the p
K
a
of the catalytic group because
pH
=
p
K
a
when
3
HA
4
=
3
A
-
4
. (See page 70 of the text.)
enzyme activity
pH
5.6
p
K
a
















