
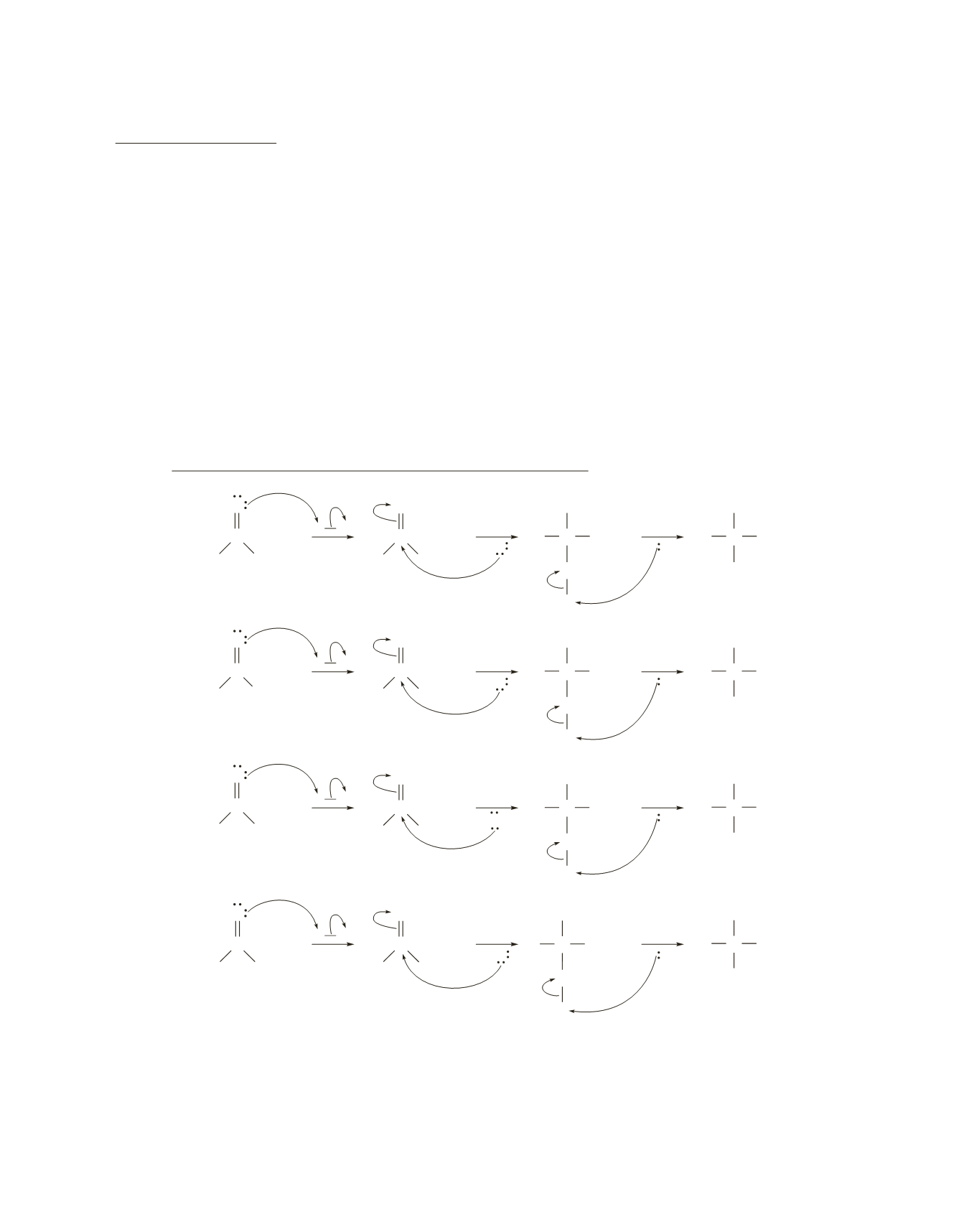
Chapter 22 737
Copyright © 2017 Pearson Education, Inc.
Solutions to Problems
1.
A catalyst increases the rate of a reaction by decreasing the difference in energy between the reactant
and the transition state of the rate-limiting step. That is, it decreases the height of the energy hill of the
rate-limiting step.
The following parameters would be different for a reaction carried out in the presence of a catalyst:
∆
H
[
,
E
a
,
∆
S
[
,
∆
G
[
,
k
rate
. These are the parameters that reflect the difference in energy between the reactant
and the transition state.
The other factors do not change because they reflect the difference in energy between the reactant and
product, which is not affected by catalysis.
2.
Notice that
(1)
and
(2)
have only the first part of the mechanism for the acid-catalyzed hydrolysis of an
ester (because the final product of the reaction is a tetrahedral compound),
(3)
has only the second part
(because the initial reactant is a tetrahedral compound), and
(4)
has both the first and second parts.
Mechanism for acid-catalyzed ester hydrolysis: first part
R C H
OH
OR
H
R C R
OH
H
R
C
OCH
3
O
O
H B
+
R C OCH
3
OH
R C OCH
3
OH
H
2
O
+
OH
OH
R C R
OH
OH
R C H
OH
ROH
OR
R C NHCH
3
OH
OH HB
+
+
(1)
(2)
(4)
H
2
HB
+
+
HB
+
+
HB
+
+
H
R C NHCH
3
OH
H
+
OH
+
OH
+
R
C
R
R
C
H
O
R
C
NHCH
3
O
+
R
C
OCH
3
OH
+
R
C
R
OH
+
R
C
H
OH
+
R
C
NHCH
3
OH
H B
+
H B
+
+
H B
B
B
B
B
H
2
O
O
a. Similarities:
the first step is protonation of the carbonyl compound, the second step is addition of a
nucleophile to the protonated carbonyl compound, and the third step is loss of a proton.
b. Differences:
the carbonyl compound that is used as the starting material; the nucleophile used in
(2)
is
an alcohol rather than water.
















