
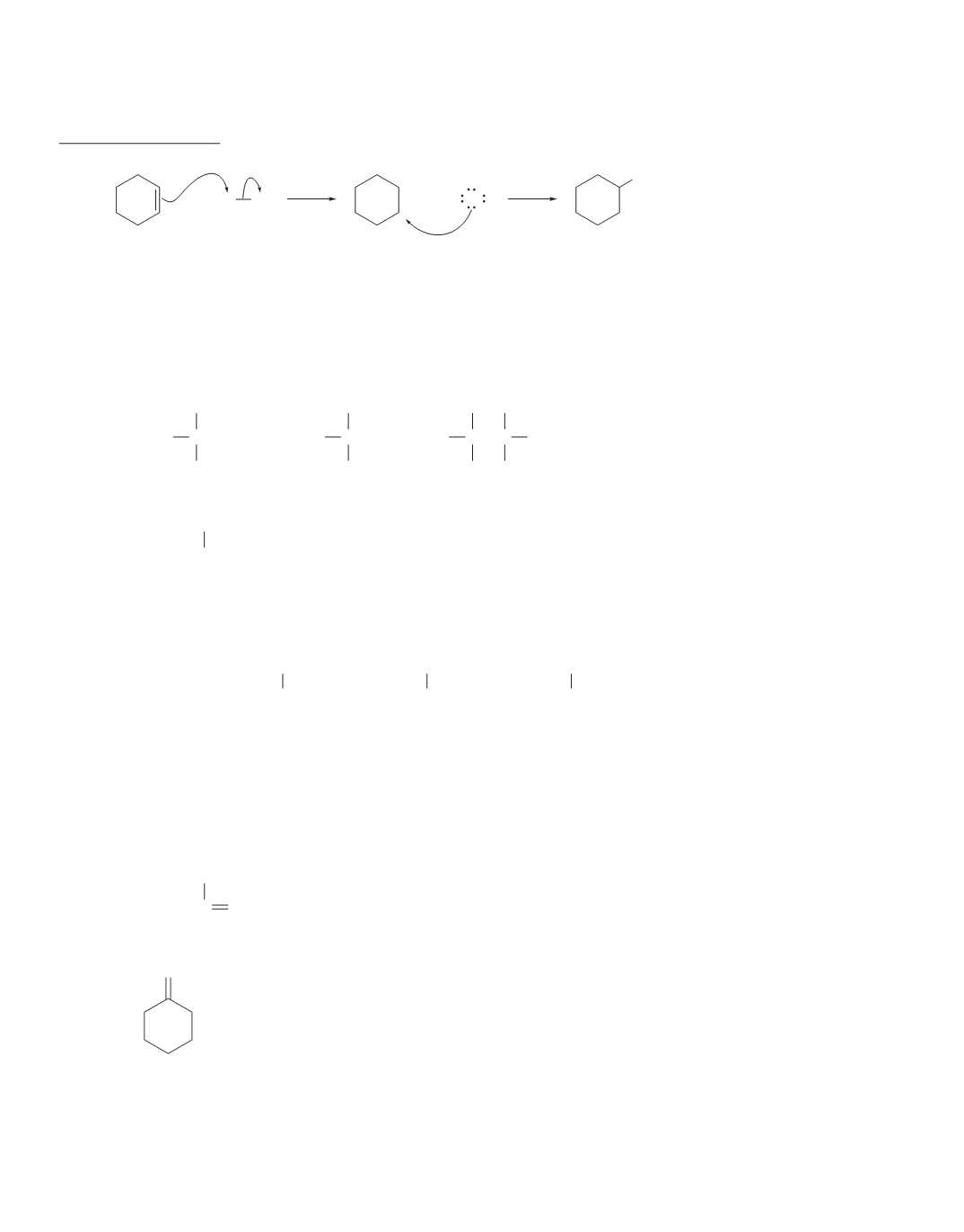
210 Chapter 6
Copyright © 2017 Pearson Education, Inc.
Solutions to Problems
1.
+
+
Cl
−
Cl
H
+
Cl
2.
a.
The
s
bond orbitals of the carbon adjacent to the positively charged carbon are available for overlap
with the vacant
p
orbital. Because the methyl cation does not have a carbon adjacent to the positively
charged carbon, no
s
bond orbitals are available for overlap with the vacant
p
orbital.
b.
An ethyl cation is more stable because the carbon adjacent to the positively charged carbon has three
s
bond orbitals available for overlap with the vacant
p
orbital, whereas a methyl cation does not have any
s
bond orbitals available for overlap with the vacant
p
orbital.
3.
a
.
CH
3
+
CCH
2
CH
3
H
CH
3
CH
2
+
CCH
2
H
H
CH
3
+
CCHC
H
H
H
H
H
3
3
6
b.
sec
-butyl cation
4.
a.
CH
3
CH
2
CCH
3
CH
3
+
CH
3
CH
2
CHCH
3
+
CH
3
CH
2
CH
2
CH
2
+
>
>
b.
The reason a halogen atom decreases the stability of the carbocation is because it is an electronegative
atom and, therefore, withdraws electrons away from the positively charged carbon. This increases the
concentration of positive charge on the carbocation, which makes it less stable.
CH
3
CHCH
2
CH
2
+
>
>
CH
3
CH
3
CHCH
2
CH
2
+
Cl
CH
3
CHCH
2
CH
2
+
F
Because fluorine is more electronegative than chlorine and, therefore, withdraws electrons more
strongly, the fluorine-substituted carbocation is less stable than the chlorine-substituted carbocation.
5.
The transition state resembles the one (reactants or products) that it is closer to on the reaction coordinate
diagram; that is, the one that it is closer to in energy.
a.
products
b.
reactants
c.
reactants
d.
products
6.
a.
CH
3
CH
2
C CH
2
CH
3
In both
a
and
b
, the compound that is more highly regioselective is the one where
the choice is between forming a tertiary carbocation and a primary carbocation.
b.
CH
2
In both
a
and
b
, the less regioselective compound is the one in which the choice
is between forming a tertiary carbocation and a secondary carbocation, because
the difference in the stability of the two possible carbocations, and therefore the
difference in the amount of product formed, is not as great as when the choice is
between a tertiary and a primary carbocation.
















