
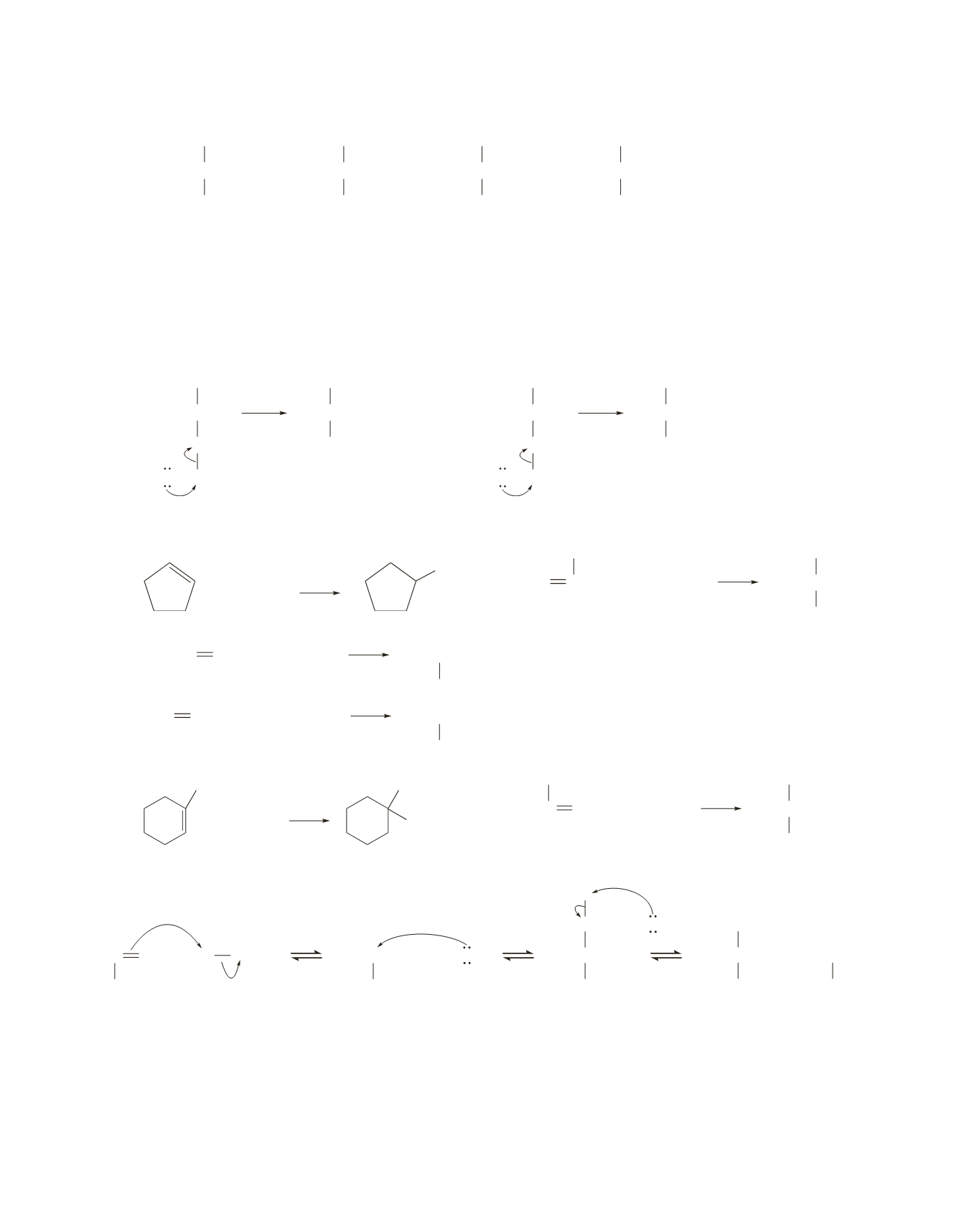
212 Chapter 6
Copyright © 2017 Pearson Education, Inc.
12.
a.
CH
3
CCH
3
Cl
CH
3
CH
3
CCH
3
Br
CH
3
CH
3
CCH
3
OH
CH
3
CH
3
CCH
3
OCH
3
CH
3
1.
2.
3.
4.
b. 1.
The first step in all the reactions is addition of an electrophilic proton
1
H
+
2
to the carbon of the
CH
2
group.
2.
A
tert
-butyl carbocation is formed as an intermediate in each of the reactions.
c. 1.
The nucleophile that adds to the
tert
-butyl carbocation is different in each reaction.
2.
In reactions #3 and #4, there is a third step—a proton is lost from the group that was the nucleo-
phile in the second step of the reaction.
CH
3
CCH
3
+
OH
CH
3
CH
3
CCH
3
OH
CH
3
CH
3
CCH
3
OCH
3
CH
3
3.
4.
H
+
H
3
O
+
+
CH
3
CCH
3
+
OCH
3
CH
3
H
H
2
O
CH
3
OH
CH
3
OH
2
+
13.
Solved in the text.
14.
a.
+
H
2
O
H
2
SO
4
OH
b.
CH
2
CCH
3
CH
3
+
CH
3
OH
CH
3
OCCH
3
H
2
SO
4
CH
3
CH
3
c.
CH
3
CH CHCH
3
+
H
2
O
CH
3
CHCH
2
CH
3
H
2
SO
4
CH
2
CHCH
2
CH
3
+
H
2
O
CH
3
CHCH
2
CH
3
H
2
SO
4
or
OH
OH
d.
+
CH
3
OH
H
2
SO
4
CH
3
3
CH
3
OCH
e.
CH
3
CHCH
3
+
H
2
O
CH
3
CCH
2
H
2
SO
4
CH
3
OH
CH
3
C
CH
3
15.
+
+
CH
3
C CH
2
CH
3
+
CH
3
CCH
3
CH
3
OSO
3
H
CH
3
CCH
3
+
OR
H
CH
3
+
+
ROH
H
CH
3
CCH
3
OR
CH
3
ROH
ROH
H
















