
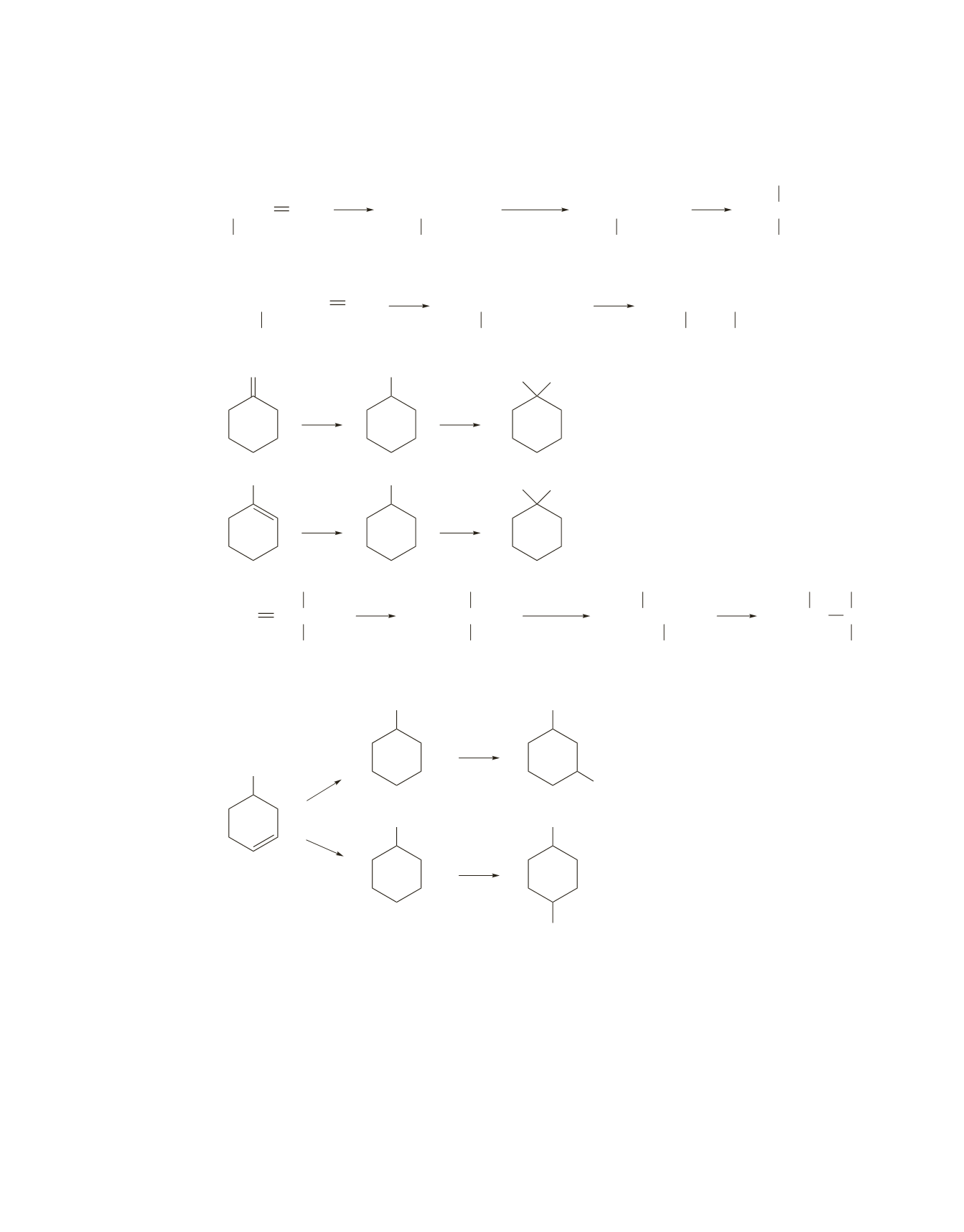
Chapter 6 213
Copyright © 2017 Pearson Education, Inc.
16.
Solved in the text.
17.
a.
CH
3
CHCH
CH
3
HBr
CH
2
CH
3
CHCHCH
3
CH
3
+
CH
3
CCH
2
CH
3
CH
3
+
Br
−
CH
3
CCH
2
CH
3
CH
3
Br
1,2-hydride
shift
secondary
tertiary
b.
CH
3
CHCH
2
CH CH
2
CH
3
CH
3
CHCH
2
CHCH
3
CH
3
+
HBr
CH
3
CHCH
2
CHCH
3
CH
3
Br
Br
−
c.
CH
2
CH
3
CH
3
Br
HBr
Br
−
+
d.
CH
3
CH
3
CH
3
Br
HBr
Br
−
+
e.
CH
2
CHCCH
3
CH
3
CH
3
HBr
Br
−
1,2-methyl
shift
tertiary carbocation
CH
3
CHCCH
3
CH
3
CH
3
+
CH
3
CHCCH
3
CH
3
CH
3
CH
3
CH
CH
3
CCH
3
Br
CH
3
secondary carbocation
+
f.
CH
3
CH
3
HBr
Br
−
+
CH
3
CH
3
CH
3
Br
Br
+
HBr
Br
−
Approximately 1-bromo-3-methylcyclohexane and 1-bromo-4-methylcyclohexane are obtained because in
each case, the intermediate is a secondary carbocation. Therefore, the two compounds are formed at about
the same rate. A carbocation rearrangement does not occur because it would just form another secondary
carbocation.
18.
The reaction with 9-BBN is more highly regioselective. 9-BBN is sterically hindered, so it will be more
likely than BH
3
to add to the least sterically hindered carbon.
















