
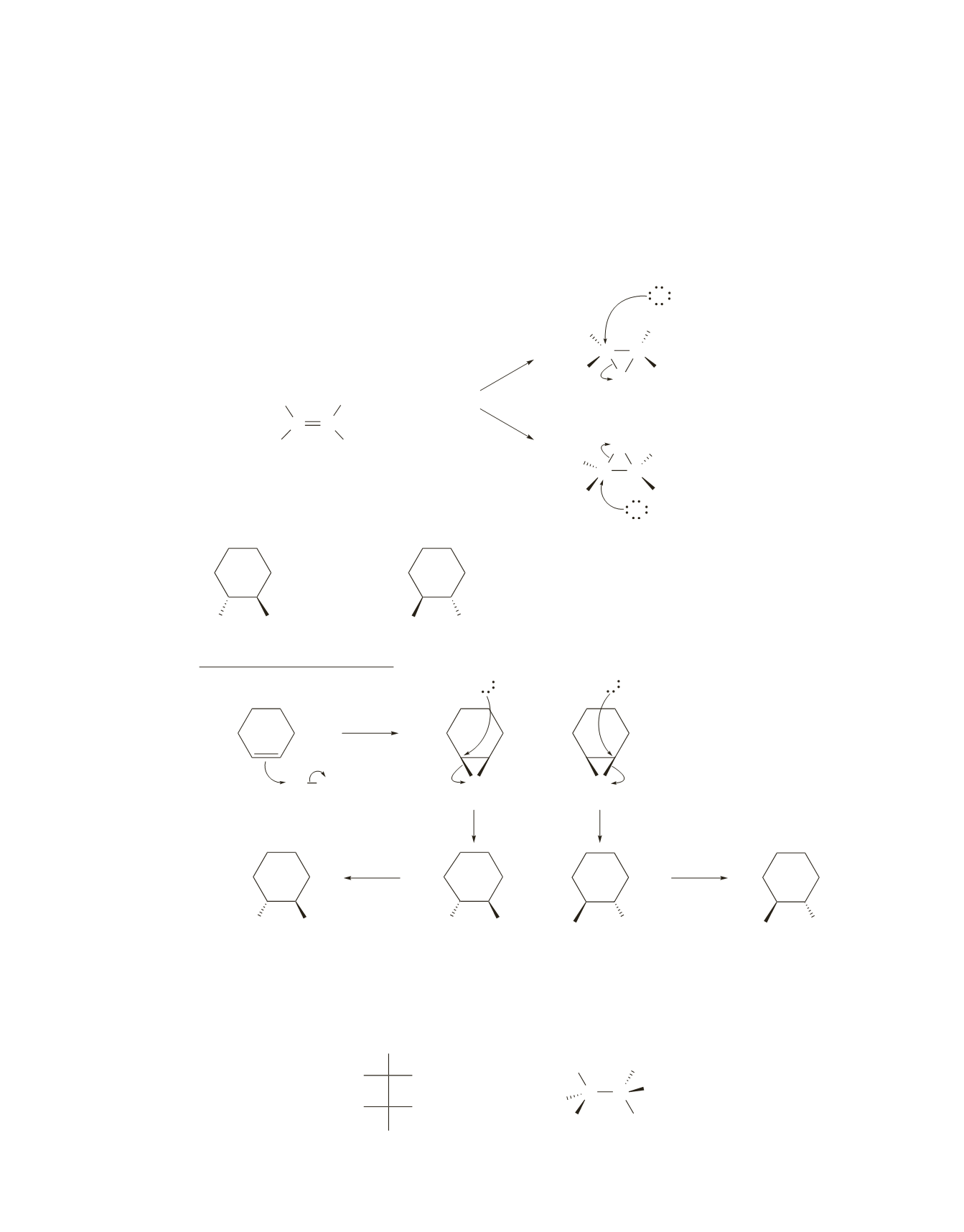
Chapter 6 219
Copyright © 2017 Pearson Education, Inc.
46.
Two different bromonium ions are formed because Br
2
can add to the double bond either from the top of the
plane or from the bottom of the plane defined by the double bond; the two bromonium ions are formed in
equal amounts. Attacking the less hindered carbon of one bromonium ion forms one stereoisomer, whereas
attacking the less hindered carbon of the other bromonium ion forms the other stereoisomer. Because Br
-
can attack the least sterically hindered carbon with equal ease from pathway
a
as from pathway
b
, equal
amounts of the threo enantiomers are obtained. Of course, some reaction will occur at the more hindered
end of the bromonium ion, but it will occur to the same extent in both pathways.
C C
H
3
C
CH
2
CH
2
CH
2
CH
3
H
H
Br
pathway
b
pathway
a
2
C C
Br
H
CH
2
CH
2
CH
2
CH
3
H
H
3
C
+
Br
C C
CH
2
CH
2
CH
2
H
H
H
3
C
+
Br
−
Br
−
Br
2
CH
3
47.
a.
and
HO Br
Br
OH
The addition of Br and OH are anti, so
these two substituents are trans to each
other in the cyclic product.
b. mechanism of the reaction
Br
Br
Br Br
H
2
O
H
2
O
Br
−
+
H
+
+
+
+
+
H
+
HO Br
Br
Br
Br
OH
OH
2
H
2
O
+
+
48.
a.
Only anti addition occurs. Because the reactant is trans, the product is expected to be the erythro pair
of enantiomers. However, in this case, each asymmetric carbon is attached to the same four groups, so
the product is a meso compound. Therefore, only one stereoisomer is obtained.
CH
2
CH
3
CH
3
Br
CH
2
CH
3
CH
3
Br
C C
CH
3
CH
2
Br
CH
2
CH
3
Br
CH
3
CH
3
S
R
S R
or
















