
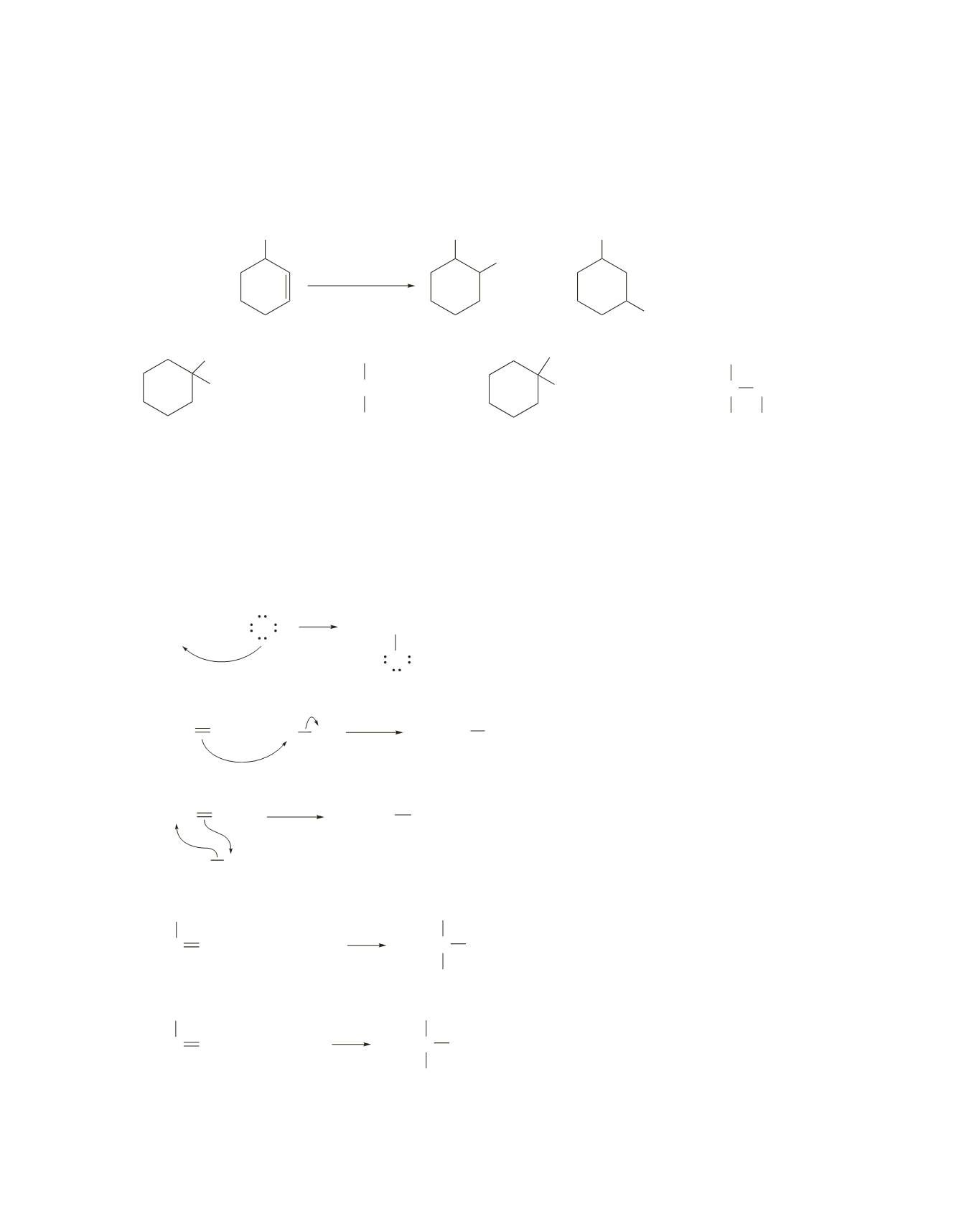
222 Chapter 6
Copyright © 2017 Pearson Education, Inc.
54.
Less of the desired product would be formed from 3-methylcyclohexene than from 1-methylcyclohexene.
Hydroboration–oxidation of 3-methylcyclohexene would form both 2-methylcyclohexanol (the desired
product) and 3-methylcyclohexanol at approximately the same rate because the transition states formed
when borane adds to the 1-position or the 2-position of the alkene have approximately the same stability.
CH
3
CH
3
OH
+
1. R BH/THF
2
CH
3
OH
2. H
2
O
2
, HO
−
, H
2
O
55.
a.
Br
CH
2
CH
3
b.
CH
3
CCH
2
CH
3
Br
CH
3
c.
CH
2
CH
3
Br
d.
CH
3
CH
2
C CHCH
3
CH
3
Br CH
3
56.
a.
addition of a hydrogen halide, acid-catalyzed addition of water, acid-catalyzed addition of an alcohol
b.
hydroboration, epoxidation
c.
addition of Br
2
or Cl
2
d.
ozonolysis
57.
a.
electrophile
CH
3
CHCH
3
+
nucleophile
Cl
−
+
CH
3
CHCH
3
Cl
b.
CH
3
CH CH
2
+
H Br
CH
3
CH CH
3
+
Br
nucleophile
electrophile
−
+
c.
CH
3
CH CH
2
+
H BH
2
CH
3
CH
2
CH
2
BH
2
electrophile nucleophile
nucleophile electrophile
58.
a.
CH
3
C
CH
3
Br
CH
2
CH
3
CH
3
C
CH
3
CHCH
3
+
HBr
b.
CH
3
C
CH
3
I
CH
2
CH
3
CH
3
C
CH
3
CHCH
3
HI
+
















