
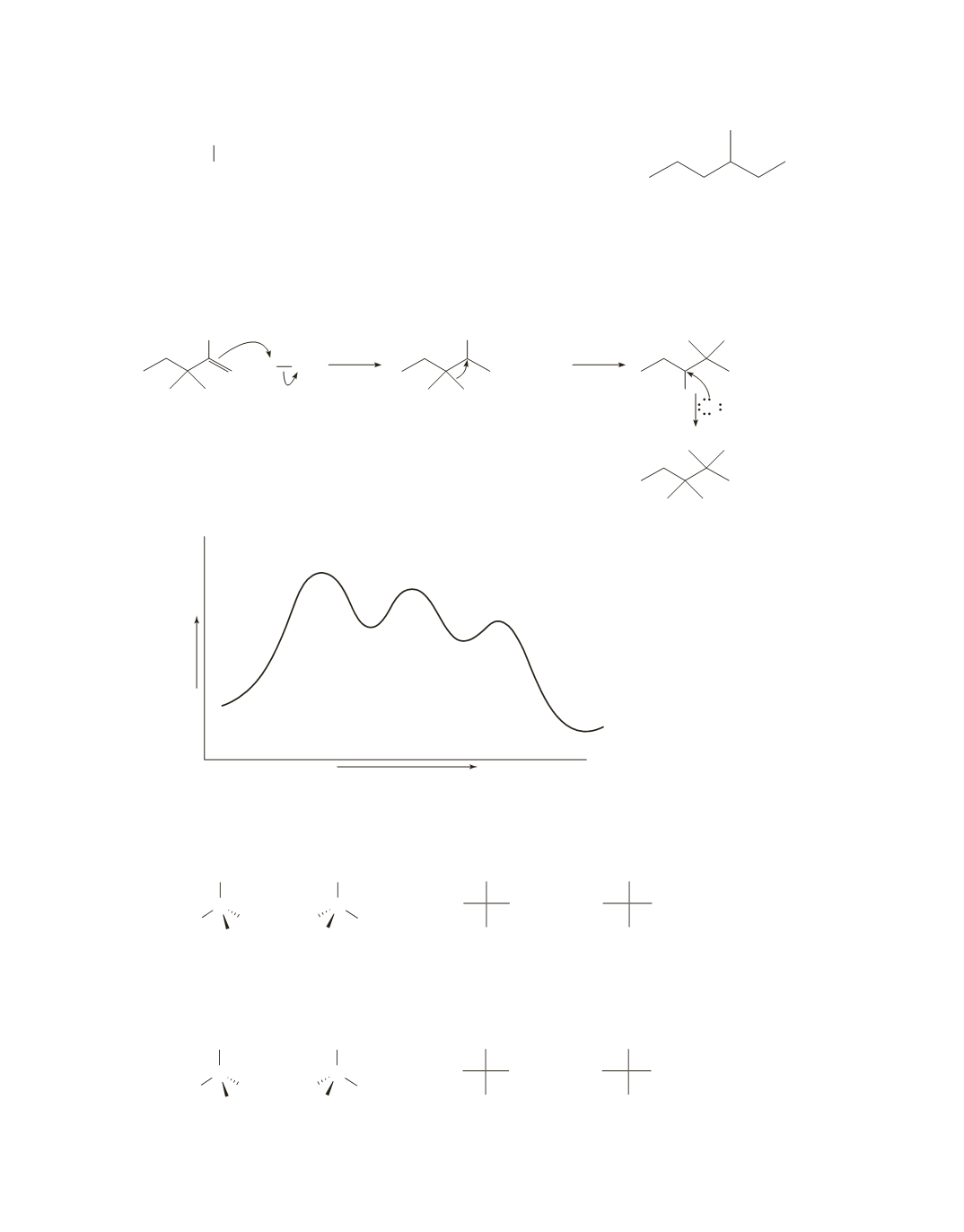
226 Chapter 6
Copyright © 2017 Pearson Education, Inc.
69.
a.
+
H
H
+
H Br
+
H
H
Br
−
+
H H
H H
Br
Br
−
b.
Progress of the reaction
Free energy
70.
a. 1.
Both
cis
- and
trans
-2-butene form these products; in each case, a product with one asymmetric
center is formed, so the product is a racemic mixture.
CH
2
CH
3
C
H CH
3
Cl
CH
2
CH
3
C
CH
3
H
Cl
+
CH
3
Cl
H
CH
2
CH
3
CH
3
H
Cl
CH
2
CH
3
+
or
S
R
S
R
2.
Both
cis
- and
trans
-2-butene form these products; in each case, a product with one asymmetric
center is formed, so the product is a racemic mixture.
CH
2
CH
3
C
H
OH
CH
2
CH
3
C
CH
3
H
HO
+
CH
3
OH
H
CH
2
CH
3
CH
3
H
HO
CH
2
CH
3
+
or
S
R
S
R
CH
3
68.
a.
CH
3
CCH
3
CH
3
+
Tertiary is more stable
than secondary.
b.
CH
3
CHCH
3
+
The electron–withdrawing
chlorine destabilizes the carbocation
by increasing the amount of positive
charge on the carbon.
c.
+
Tertiary is more stable
than secondary.
















