
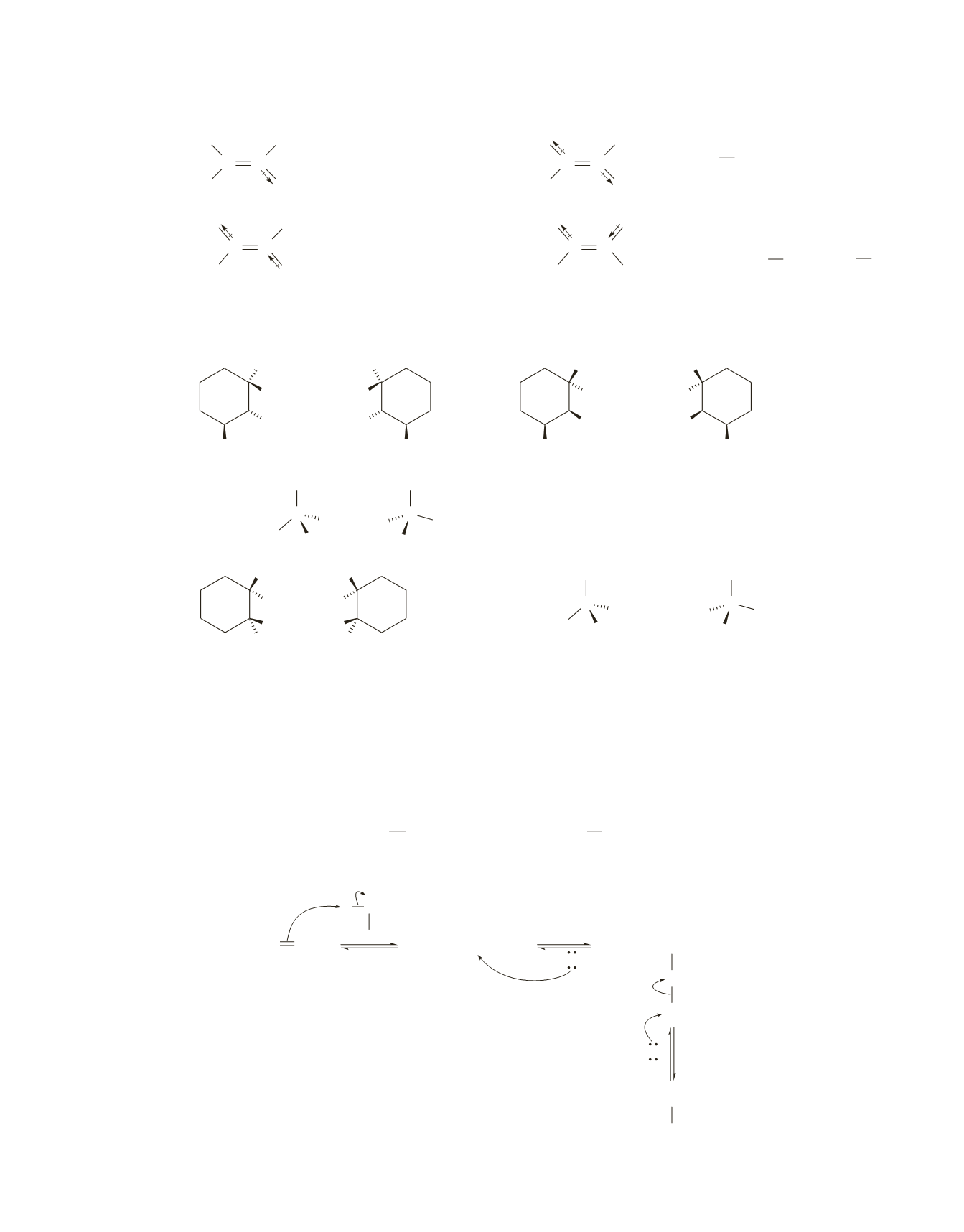
Chapter 6 229
Copyright © 2017 Pearson Education, Inc.
73.
a.
C C
H
H
Cl
H
has a greater dipole moment than
C C
H
H
Cl
Cl
where the C Cl dipoles
oppose each other
b.
C C
H
H
CH
3
Cl
has a greater dipole moment than
Recall that an
sp
2
carbon is more
electronegative than an
sp
3
carbon.
Therefore, the C CH
3
and C Cl
dipoles reinforce each other, leading to a
higher dipole, in the compound on
the left and oppose each other in the
compound on the right.
C C
H
CH
3
H
Cl
74.
a.
CH
2
CH
3
+
Br
Br
CH
3
Br
Br
CH
3
Br
CH
2
CH
3
CH
2
CH
3
+
+
Br
CH
3
Br
Br
CH
3
CH
2
CH
3
b.
CH
3
CH
3
CH
2
CH
2
OH
H H
CH
3
HO
CH
2
CH
2
CH
3
+
C
C
c.
Br
Br
CH
3
CH
3
CH
3
Br
Br
CH
3
d.
CH
3
CH
3
CH
2
CH
2
Br
CH
3
CH
2
CH
3
CH
2
CH
3
Br
CH
2
CH
2
CH
3
+
C
C
75.
No, he should not follow his friend’s advice. Adding the electrophile to the
sp
2
carbon bonded to the most
hydrogens forms a secondary carbocation in preference to a primary carbocation. However, in this case,
the primary carbocation is more stable than the secondary carbocation. The electron-withdrawing fluorine
substituents are closer to the positively charged carbon in the secondary carbon. Therefore, they will desta-
bilize the secondary carbocation more than the primary carbocation. So the major product will be 1,1,1-tri-
fluoro-3-iodopropane and not 1,1,1-trifluoro-2-iodopropane, the compound that would be predicted to be
the major product by following the rule.
F
3
CCH
2
CH
2
+
more stable
F
3
CCH CH
3
+
less stable because of the nearby
electron-withdrawing uorines
76.
a.
CH
3
CH
2
CH CH
2
CH
3
CH
2
CHCH
3
+
+
CH
3
OH
CH
3
CH
2
CHCH
3
OCH
3
H
CH
3
OH
CH
3
CH
2
CHCH
3
OCH
3
+
H
H OCH
3
















