
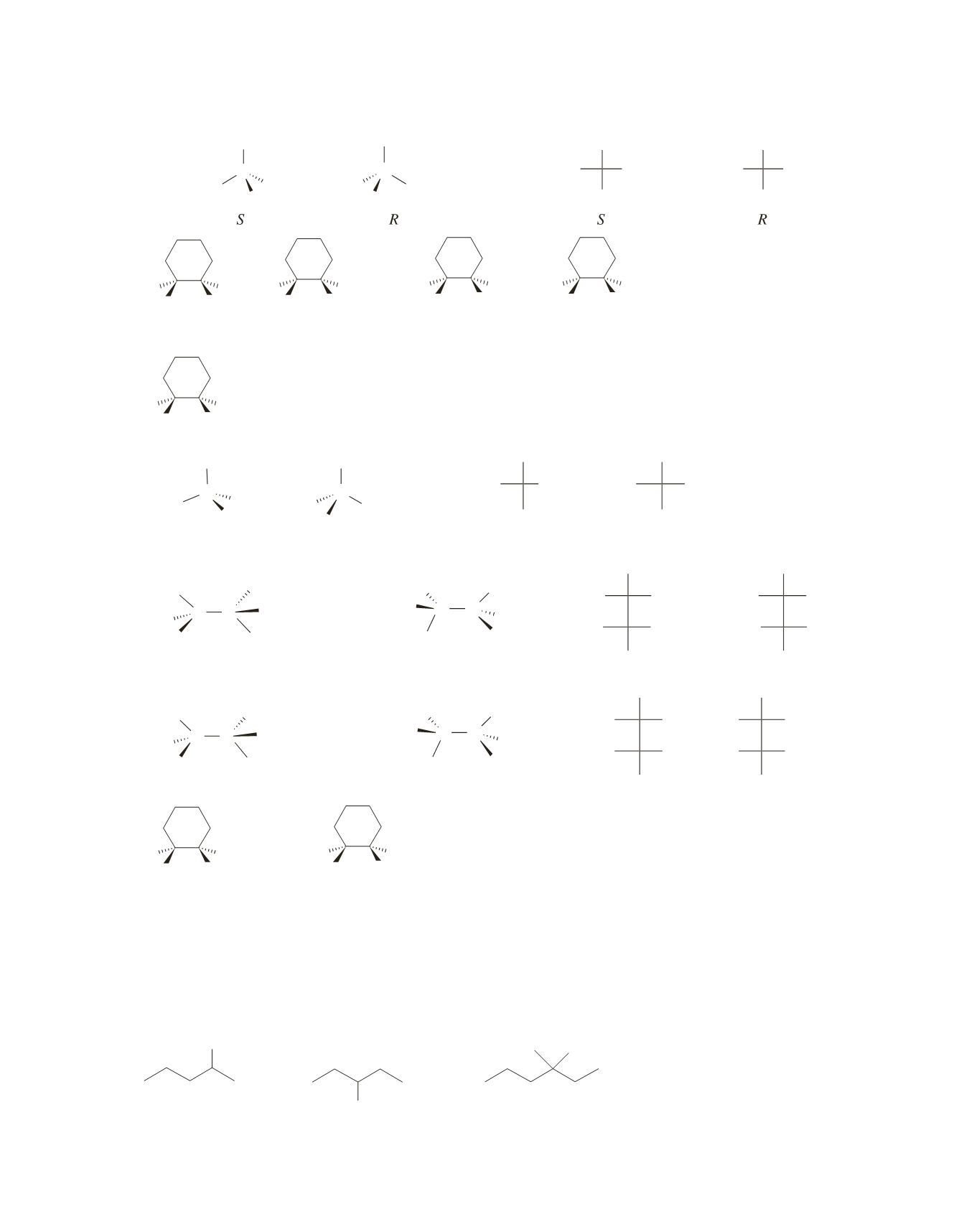
232 Chapter 6
Copyright © 2017 Pearson Education, Inc.
d.
CH(CH
3
)
2
CH
3
H
CH
2
CH
2
CH
3
CH(CH
3
)
2
H CH
3
CH
2
CH
2
CH
3
or
CH(CH
3
)
2
H
CH
3
CH
3
CH
3
CH
2
CH
2
CH(CH
3
)
2
H CH
2
CH
2
CH
3
C
+
+
C
e.
CH
3
CH
3
CH
3
CH
3
CH
3
H
Cl
+
+
+
CH
3
Cl
H
Cl
H
CH
3
Cl
CH
3
H
cis
trans
R
S
S
S
R
R
R S
f.
D D
H
H
S R
g.
(CH
3
)
2
CCH
2
CH
3
(CH
3
)
2
CCH
2
CH
3
(CH
3
)
2
CCH
2
CH
3
(CH
3
)
2
CCH
2
CH
3
C H
Br
C
Br
CH
2
Br
H
or
CH
2
Br
Br
H
CH
2
Br
H
Br
R
S
R
S
BrCH
2
h.
C C CH
3
CH
3
CH
3
H
CH
2
CH
2
CH
3
H
CH
3
CH
2
+
C C
H
CH
2
CH
3
H
CH
3
or
CH
2
CH
3
CH
2
CH
2
CH
3
H
CH
3
CH
3
H
CH
2
CH
3
CH
2
CH
2
CH
3
CH
3
H
H
CH
3
+
S S
R R
S
S
R
R
CH
3
CH
2
CH
2
i.
C C CH
3
CH
3
CH
3
H
CH
2
CH
2
CH
3
H
CH
3
CH
2
+
C C
H
CH
2
CH
3
CH
3
H
or
CH
2
CH
3
CH
2
CH
2
CH
3
H
CH
3
H
CH
3
CH
2
CH
3
CH
2
CH
2
CH
3
CH
3
H
CH
3
H
+
R S
R S
R
S
S
R
CH
3
CH
2
CH
2
j.
Cl
CH
2
CH
3
H
H
Cl
H
H
CH
3
S
S
R
R
CH
2
83.
a.
Both 1-butene and 2-butene react with HCl to form 2-chlorobutane.
b.
Both alkenes form the same carbocation and, therefore, have transition states that are close in energy,
but because 2-butene is more stable than 1-butene, 2-butene has the greater free energy of activation.
c.
Both compounds form the same carbocation and, therefore, have transition states that are close in
energy, but because (
Z
)-2-butene is less stable, it reacts more rapidly with HCl.
84.
a.
Br
Br
and
b.
Br
















