
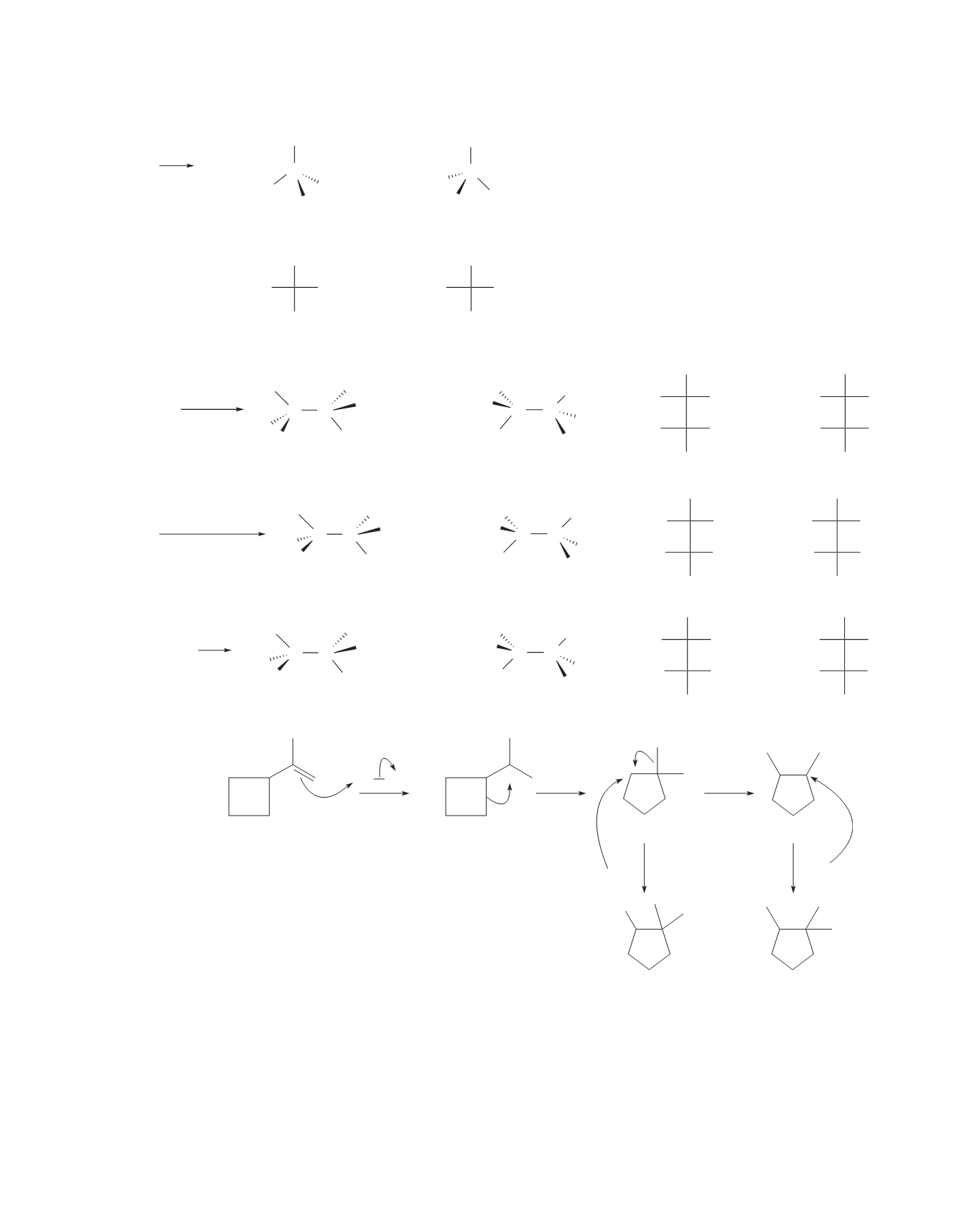
Chapter 6 237
Copyright © 2017 Pearson Education, Inc.
(CH
3
)
H
2
H
C
CH
3
CH
2
CH
3
H
C
CH(CH
3
)
3
H
3
C
+
or
Pd/C
CH
3
R
S
CH(CH
3
)
H CH
3
CH
2
CH
3
R
CH(CH
3
)
H
CH
2
CH
3
S
CH
3
CH
CH
2
(CH
3
)CH
C C
H
CH
3
CH
3
CH(CH
3
)
2
Br
Br
+
C C
H
3
C
Br
H
CH
3
Br
Br
H
CH
3
CH
3
CH(CH
3
)
2
Br
CH
3
Br
H
CH(CH
3
)
2
CH
3
Br
+
or
CH
2
Cl
2
Br
2
S S
R R
R
R
S
S
(CH
3
)CH
2. HO
−
, H
2
O
2
, H
2
O
C C
H
3
C
H
HO
CH(CH
3
)
2
H
CH
3
+
C C
H
H
3
C
CH
3
OH
H
H OH
CH(CH
3
)
2
CH
3
CH
3
CH
3
H
HO H
CH(CH
3
)
2
H CH
3
or
1. R
2
BH/THF
S S
R R
S
S
+
R
R
C C
H
CH
3
CH
3
CH(CH
3
)
2
Br
OH
+
C C
H
3
C
HO
H
CH
3
Br
H Br
CH
3
CH
3
CH(CH
3
)
2
HO CH
3
Br
H
CH(CH
3
)
2
CH
3
OH
+
or
H
2
O
Br
2
(CH
3
)CH
S S
R R
S
S
R
R
96.
a.
H Br
+
+
.. ..
..
..
Br
−
.. ..
..
..
Br
Br
Br
−
+
secondary
tertiary
b.
The initially formed carbocation is tertiary.
c.
The rearranged carbocation is secondary, which undergoes another rearrangement to a more stable ter-
tiary carbocation.
d.
The initially formed carbocation rearranges in order to release the strain in the four-membered ring.
(A tertiary carbocation with a strained four-membered ring is less stable than a secondary carbocation
with an unstrained five-membered ring.)
















