
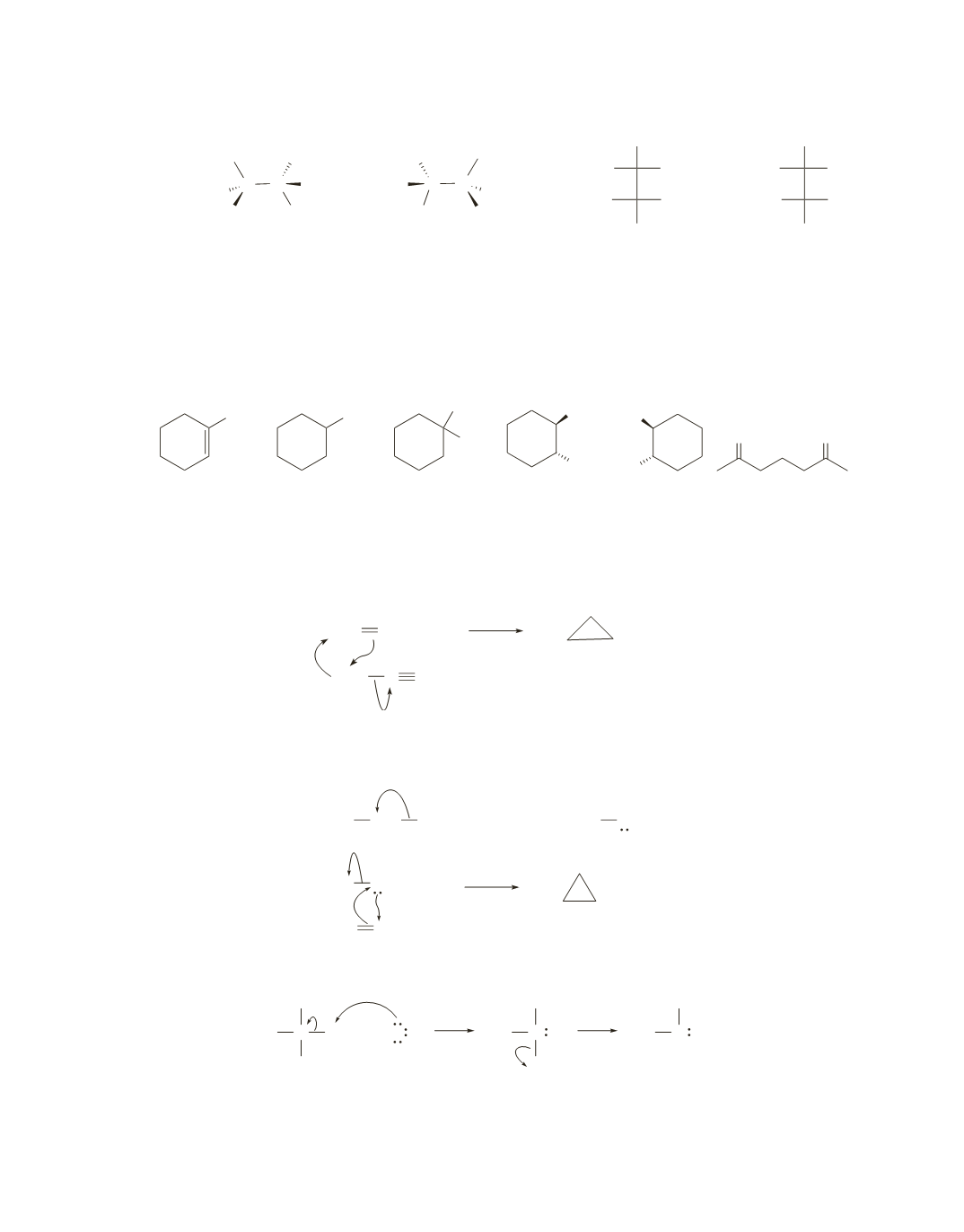
Chapter 6 235
Copyright © 2017 Pearson Education, Inc.
l.
CC
H
CH
2
CH
3
CH
3
CH
3
H
or
CH
2
CH
3
H CH
3
CH
2
CH
3
CH
3
H
C C
CH
2
CH
3
H
CH
3
CH
2
CH
3
CH
3
H
CH
2
CH
3
CH
3
H
CH
2
CH
3
H CH
3
+
CH
3
S S
R R
R
R
S
S
+
CH
2
88.
The first product would not be formed, because none of the bonds attached to C-2 were broken during the
reaction. Therefore, the configuration at C-2 cannot change.
89.
A
has two degrees of unsaturation. Because it has three methylene groups, it must be a methyl-
substituted cyclopentane. Because
A
forms only one product when it reacts with aqueous acid,
A
must be
1-methylcyclopentene. Therefore,
A–F
have the following structures.
CH
A
B
C
E
F
3
CH
3
OH
CH
3
HO
O
O
H
OH
D
CH
3
CH
3
90.
Diazomethane is a very reactive compound because the triple-bonded nitrogen has a strong propensity
to depart from the carbon to form a very stable molecule of nitrogen gas. As it departs, the nucleophilic
alkene attacks the electrophilic carbon, and in the same step, the lone pair is the nucleophile that adds to
the other
sp
2
carbon of the alkene.
+
CH
2
N N
..
_
CH
2
CH
2
+ N
2
91.
I
¬
CH
2
reacts like Br
2
. The CH
2
group is the electrophile that adds to the
sp
2
carbon, and its lone pair is
the nucleophile that adds to the other
sp
2
carbon.
CH
2
CH
2
+
–
+
I
I
–
CH
2
ZnI
reacts as if it were
–
I CH
2
I CH
2
ZnI
92.
a.
The base removes a proton. Then do what is needed to get the known product of the reaction.
C
Cl
H
+
Cl
Cl
HO
−
C
Cl
Cl
Cl
−
C Cl
Cl
+
Cl
−
+
H
2
O
−
..
















