
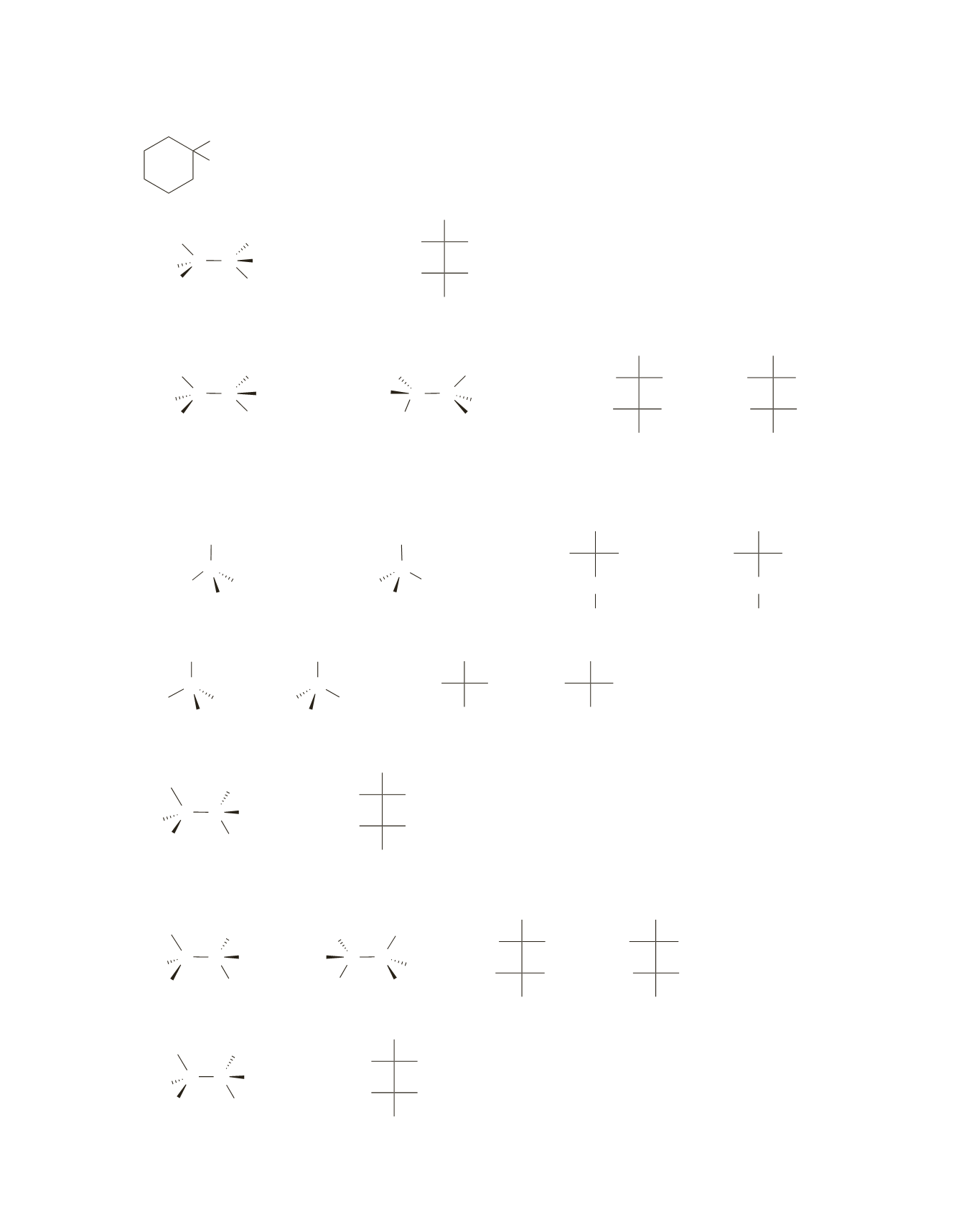
234 Chapter 6
Copyright © 2017 Pearson Education, Inc.
d.
Br
CH
3
e.
C C Br
H
CH
2
CH
3
CH
3
CH
2
H
Br
CH
2
CH
3
H Br
CH
2
CH
3
H Br
or
R S
a meso compound
S
R
f.
CH
3
CC Br
H
CH
2
CH
3
Br
H
CH
2
CH
3
H Br
CH
2
CH
3
Br
H
or
C C Br
H
CH
2
CH
3
CH
3
CH
2
Br
H
CH
2
CH
3
Br
H
CH
2
CH
3
H Br
+
S S
R R
S
S
R
R
+
CH
2
g.
The initially formed carbocation is secondary. It undergoes a 1,2-methyl shift to form a tertiary carbo-
cation that forms the products shown below.
(CH
3
)
2
Br
C
CH
2
CH
3
CH
3
Br
C
CH(CH
3
)
2
CH
3
CH
2
CH
3
or
CH
2
CH
3
CH
3
Br
CHCH
3
CH
3
CH
2
CH
3
Br
CH
3
CHCH
3
CH
3
+
+
R
S
R
S
CH
h.
CH
2
CH
3
C
H
Br
CH
2
CH
3
C
CH
3
CH
3
H
Br
+
or
CH
3
CH
2
CH
3
Br
H
CH
3
CH
2
CH
3
H
Br
+
S
R
S
R
i.
C C
CH
3
H
H
3
C
H
Cl
Cl
or
CH
3
H Cl
CH
3
H Cl
R S
S
R
a meso compound
j.
CC
CH
3
H
CH
3
Cl
Cl
H
or
CH
3
H Cl
CH
3
Cl
H
C C
CH
3
H
H
3
C
Cl
Cl
H
CH
3
Cl
H
CH
3
H Cl
+
S S
R R
S
S
+
R
R
k.
C C
CH
2
CH
3
H
CH
3
CH
2
H
CH
3
CH
3
or
CH
2
CH
3
H CH
3
CH
2
CH
3
H CH
3
R S
S
R
a meso compound
















