
228 Chapter 6
Copyright © 2017 Pearson Education, Inc.
7.
Both
cis
- and
trans
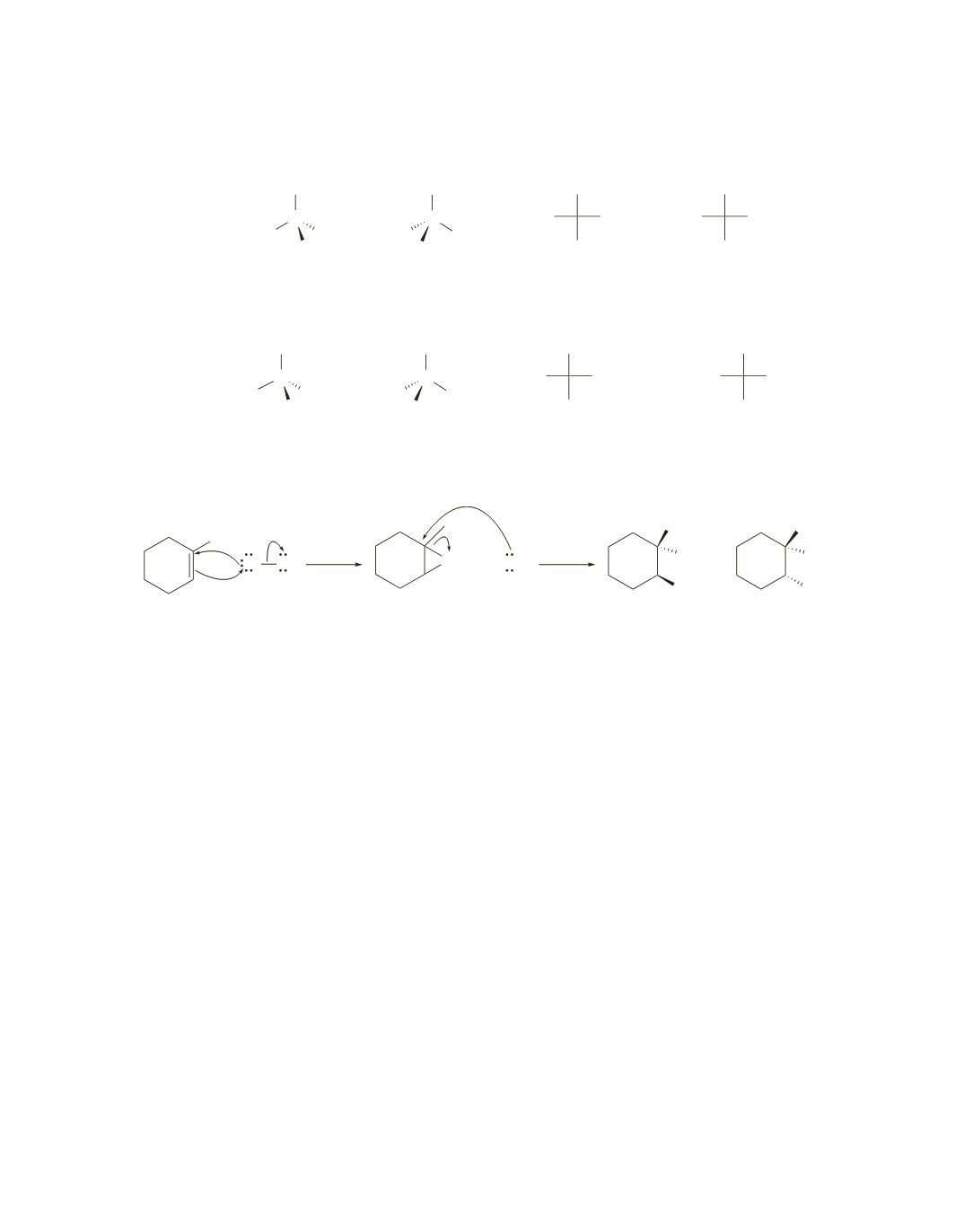
-2-butene form these products; a product with one asymmetric center is
formed, so the product is a racemic mixture.
CH
2
CH
3
C
H
OH
CH
2
CH
3
C
CH
3
H
HO
+
CH
3
OH
CH
2
CH
3
H
or
CH
3
H
CH
2
CH
3
HO
+
S
R
S
R
CH
3
8.
Both
cis
- and
trans
-2-butene form these products; a product with one asymmetric center is
formed, so the product is a racemic mixture.
CH
2
CH
3
C
H
OCH
3
CH
2
CH
3
C
CH
3
H
CH
3
O
+
CH
3
OCH
3
CH
2
CH
3
H
or
CH
3
H
CH
2
CH
3
CH
3
O
+
S
R
S
R
CH
3
b.
For
cis
- and
trans
-2-butene to form different products, (1) the reaction must form a product with two new
asymmetric centers and (2) either syn or anti addition must occur (but not both). Therefore, the
cis
- and
trans
-2-butene form different products when they react with a peroxyacid, with Br
2
, and with Br
2
in H
2
O.
71.
a.
CH
3
CH
3
Br
CH
CH
3
CH
3
3
Br
Br
Br Br
OH
OCH
3
OCH
3
+
b.
The addition of Br
2
/CH
3
OH is anti. In the second step of the reaction, CH
3
OH can add to the cyclic
bromonium ion from the top of the plane or from the bottom of the plane. The products are a pair of
enantiomers.
72.
a.
To determine relative rates, the rate constant of each alkene is divided by the smallest rate constant of
the series
1
3.51
*
10
-
8
2
.
relative rates
propene
=
1
4.95
*
10
-
8
2>1
3.51
*
10
-
8
2
=
1.41
1
Z
2
@
2
@
butene
=
1
8.32
*
10
-
8
2>1
3.51
*
10
-
8
2
=
2.37
1
E
2
@
2
@
butene
=
1
3.51
*
10
-
8
2>1
3.51
*
10
-
8
2
=
1
2
@
methyl
@
2
@
butene
=
1
2.15
*
10
-
4
2>1
3.51
*
10
-
8
2
=
6.12
*
10
3
2,3
@
dimethyl
@
2
@
butene
=
1
3.42
*
10
-
4
2>1
3.51
*
10
-
8
2
=
9.74
*
10
3
b.
Both compounds form the same carbocation but, because (
Z
)-2-butene is less stable than
E
-2-butene,
(
Z
)-2-butene has a smaller free energy of activation.
c.
2-Methyl-2-butene is more stable than (
Z
)-2-butene, and it forms a more stable carbocation intermedi-
ate (tertiary) and, therefore, a more stable transition state than does (
Z
)-2-butene (secondary). Knowing
that 2-methyl-2-butene reacts faster tells us that the energy difference between the transition states
is greater than the energy difference between the alkenes. This is what we would expect from the
Hammond postulate, because the transition states look more like the carbocations than like the alkenes.
d.
2,3-Dimethyl-2-butene is more stable than 2-methylbutene, and both compounds form a tertiary carboca-
tion intermediate. On this basis, you would predict that 2,3-dimethyl-2-butene would react more slowly
than 2-methylbutane. However, 2,3-dimethyl-2-butene has two
sp
2
carbons that can react with a proton to
form the tertiary carbocation, whereas 2-methyl-2-butene has only one. The fact that 2,3-dimethyl-2-butene
reacts faster in spite of being more stable tells us that the more important factor is the greater number of
collisions with the proper orientation that lead to a productive reaction in the case of 2,3-dimethyl-2-butene.
















