
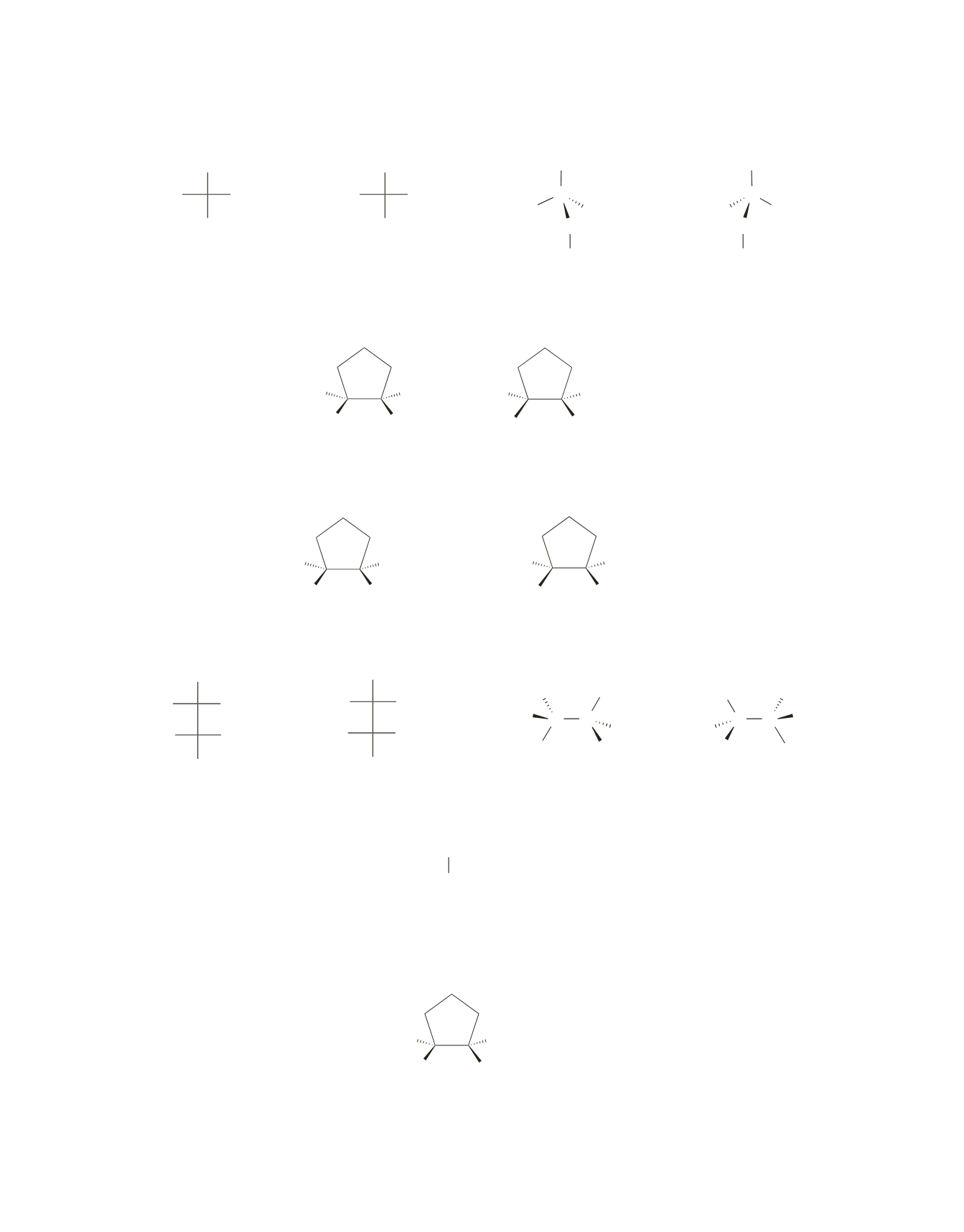
220 Chapter 6
Copyright © 2017 Pearson Education, Inc.
b.
Only one asymmetric center is created in the product, so the product is a racemic mixture.
R
R
S
S
CH
2
CH
3
BrC(CH
2
CH
3
)
2
CH
3
Br
CH
2
CH
3
BrC(CH
2
CH
3
)
2
Br
CH
3
+
CH
3
Br
C
H
C(
)
CH
2
2
CH
3
C)
(CH
3
2
CH
2
Br
C
CH
3
H
or
Br
Br
c.
Only anti addition occurs. Because the reactant is cis (a cyclopentene ring cannot exist in a trans con-
figuration), the product is the pair of enantiomers with the bromines on opposite sides of the ring.
CH
3
Br
Br
CH
3
Br
CH
3
CH
3
Br
d.
Only anti addition occurs. Because the reactant is cis, the product is the pair of enantiomers with the
bromines on opposite sides of the ring.
CH
3
Br
Br
CH
3
Br
CH
2
CH
3
CH
2
CH
3
Br
49.
a.
Only syn addition occurs. Because the reactant is trans, the product is the threo pair of enantiomers.
CH
2
CH
3
CH
3
H
CH
2
CH
3
H CH
3
CC
CH
2
CH
3
CH
3
H
H
CH
2
CH
3
CH
3
H
CH
2
CH
3
H
CH
3
C C
CH
3
CH
2
CH
2
CH
3
H
CH
3
H
+
+
or
R
R
R
S
S
R
S S
CH
3
CH
3
CH
3
CH
2
b.
The product of the reactions does not have any asymmetric centers, so it does not have any stereoisomers.
CH
3
CH CH
2
CH
2
CH
3
CH
3
c.
Only syn addition occurs. Because the reactant is cis, the product would be the pair of enantiomers
with the hydrogens on the same side of the ring, but in this case, the product is a meso compound, so
only one stereoisomer is obtained.
CH
3
CH
3
H
H
















