
Chapter 5 205
Copyright © 2017 Pearson Education, Inc.
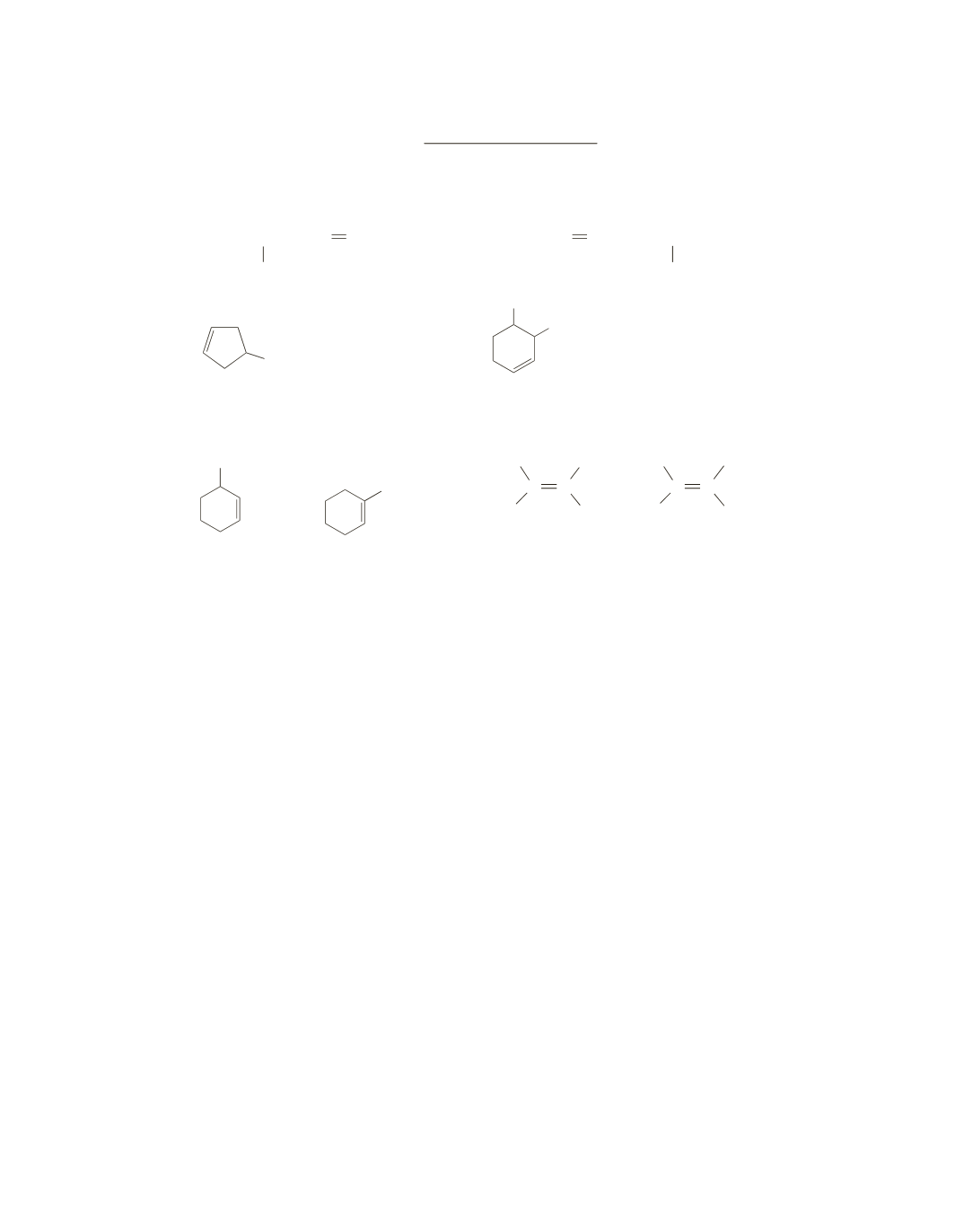
Chapter 5 Practice Test
1.
Name each of the following:
a.
CH
3
CH
2
CHCH
2
CH CH
2
CH
3
b.
Br
c.
CH
3
CH
2
CH CHCH
2
CH
2
CHCH
3
CH
2
CH
3
d.
Cl
CH
3
2.
Which member of each pair is more stable?
a.
CH
3
CH
3
or
b.
C C
H
3
C
H
CH
3
H
or
C C
H
3
C
H
H
CH
3
3.
Correct the incorrect names.
a.
3-pentene
c.
2-ethyl-2-butene
b.
2-vinylpentane
d.
2-methylcyclohexene
4.
Indicate whether each of the following statements is true or false:
a.
Increasing the energy of activation increases the rate of the reaction.
T F
b.
Decreasing the entropy of the products compared to the entropy of the
reactants makes the equilibrium constant more favorable.
T F
c.
An exergonic reaction is one with a
-
∆
G
°
.
T F
d.
An alkene is an electrophile.
T F
e.
The higher the energy of activation, the more slowly the reaction takes place.
T F
f.
Another name for
trans
-2-butene is
1
Z
2
-2-butene.
T F
g.
A reaction with a negative
∆
G
°
has an equilibrium constant greater than one.
T F
h.
Increasing the free energy of the reactants increases the rate of the reaction.
T F
i.
Increasing the free energy of the products increases the rate of the reaction.
T F
j.
The magnitude of a rate constant is not dependent on the concentration of the reactants.
T F
k.
2,3-Dimethyl-2-pentene is more stable than 3,4-dimethyl-2-pentene.
T F
















