
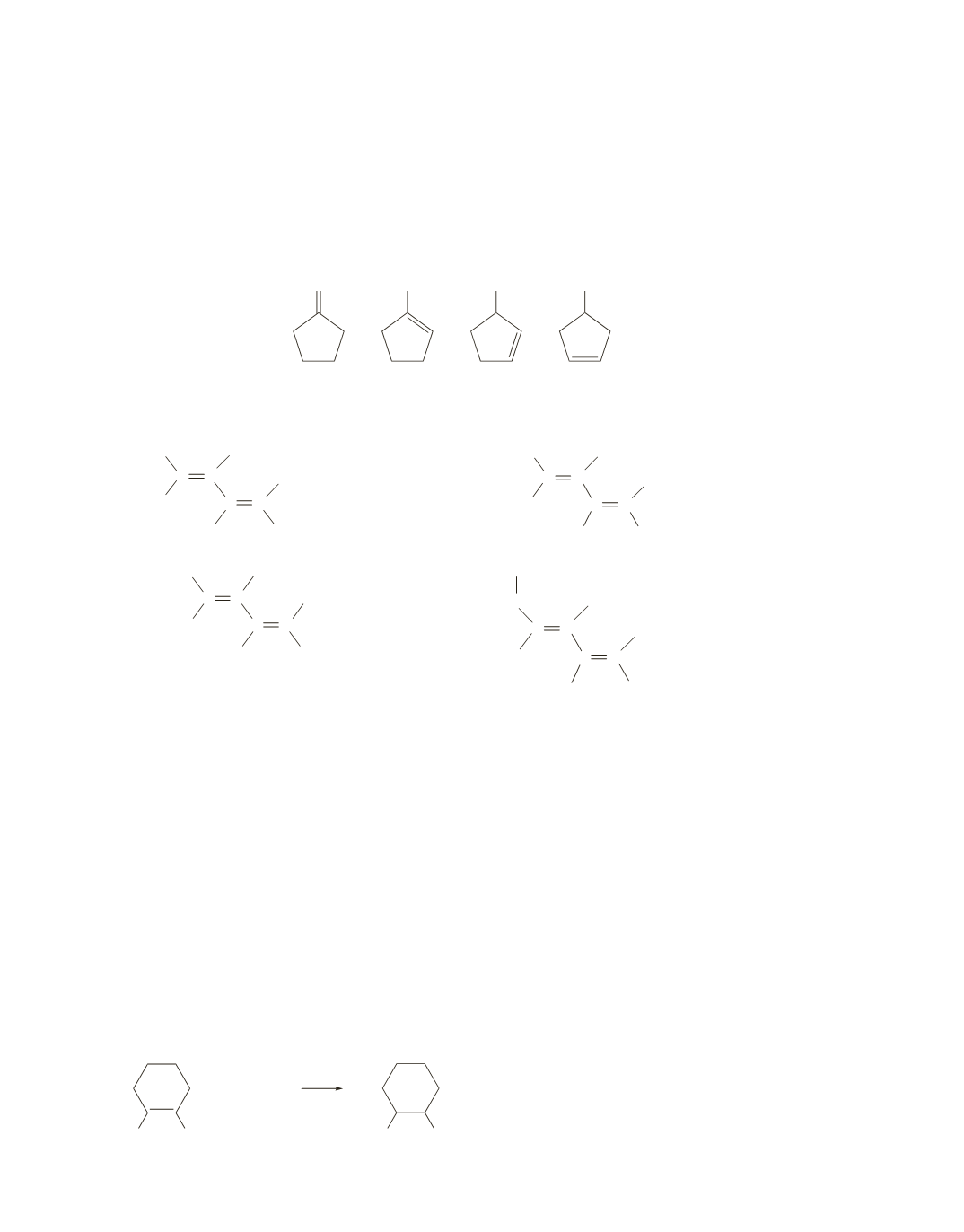
202 Chapter 5
Copyright © 2017 Pearson Education, Inc.
d.
3-heptene
e.
4-ethylcyclohexene
f.
2-chloro-3-hexene
g.
3-methyl-2-pentene
h.
2-methyl-1-hexene (it does not have
E
,
Z
isomers)
i.
1-methylcyclopentene
56.
a.
4 alkenes
CH
2
CH
3
CH
3
CH
3
b.
1-Methylcyclopentene is the most stable.
c.
Because 1-methylcyclopentene is the most stable, it has the smallest heat of hydrogenation.
57.
a.
H
C C
C
CH
3
H
CH
3
C
H
ClCH
2
b.
C
H
C
C
CH
3
H CH
2
CH
2
CH
3
C
H
CH
3
CH
2
c.
C
H
C
C
H
C
CH
2
CH
2
CH
3
CH
3
CH
2
CH
3
CH
3
d.
H
C C
C
H
Br
H
C
CH
2
CH
3
CH
3
CH
Br
58.
a.
2
b.
B, D, F
c.
E to G (The fastest step has the smallest energy of activation to overcome.)
d.
G
e.
A
f.
C
g.
C
h.
endergonic
i.
exergonic
j.
E to G (The largest rate constant corresponds to the smallest energy of activation.)
k.
G to E (The smallest rate constant corresponds to the largest energy of activation.)
59.
a.
B will have the larger
∆
S
°
value because, unlike A, the number of reactants is not the same as the
number of products.
b.
∆
S
°
=
1
the freedom of motion of the products
2
-
1
the freedom of motion of the reactants
2
.
Because the three products have a greater freedom of motion than the two reactants,
∆
S
°
is positive.
60.
+
CH
3
CH
3
H
2
Pd
/
C
CH
3
CH
3
















