
Chapter 5 197
Copyright © 2017 Pearson Education, Inc.
36.
a.
The first step in the forward direction
1
A
S
B
2
has the greatest free energy of activation.
b.
The first-formed intermediate
1
B
2
is more apt to revert to reactants, because the free energy of
activation for
B
to form
A
(the reactants) is less than the free energy of activation for
B
to form
C
.
c.
The second step
1
B
S
C
2
is the rate-determining step because it has the transition state with the
highest energy.
Notice that the second step is rate-determining even though the first step has the greater energy of acti-
vation (steeper hill to climb). That is because it is easier for the intermediate that is formed in the first
step to go back to starting material than to undergo the second step of the reaction. So the second step
is the rate-limiting step.
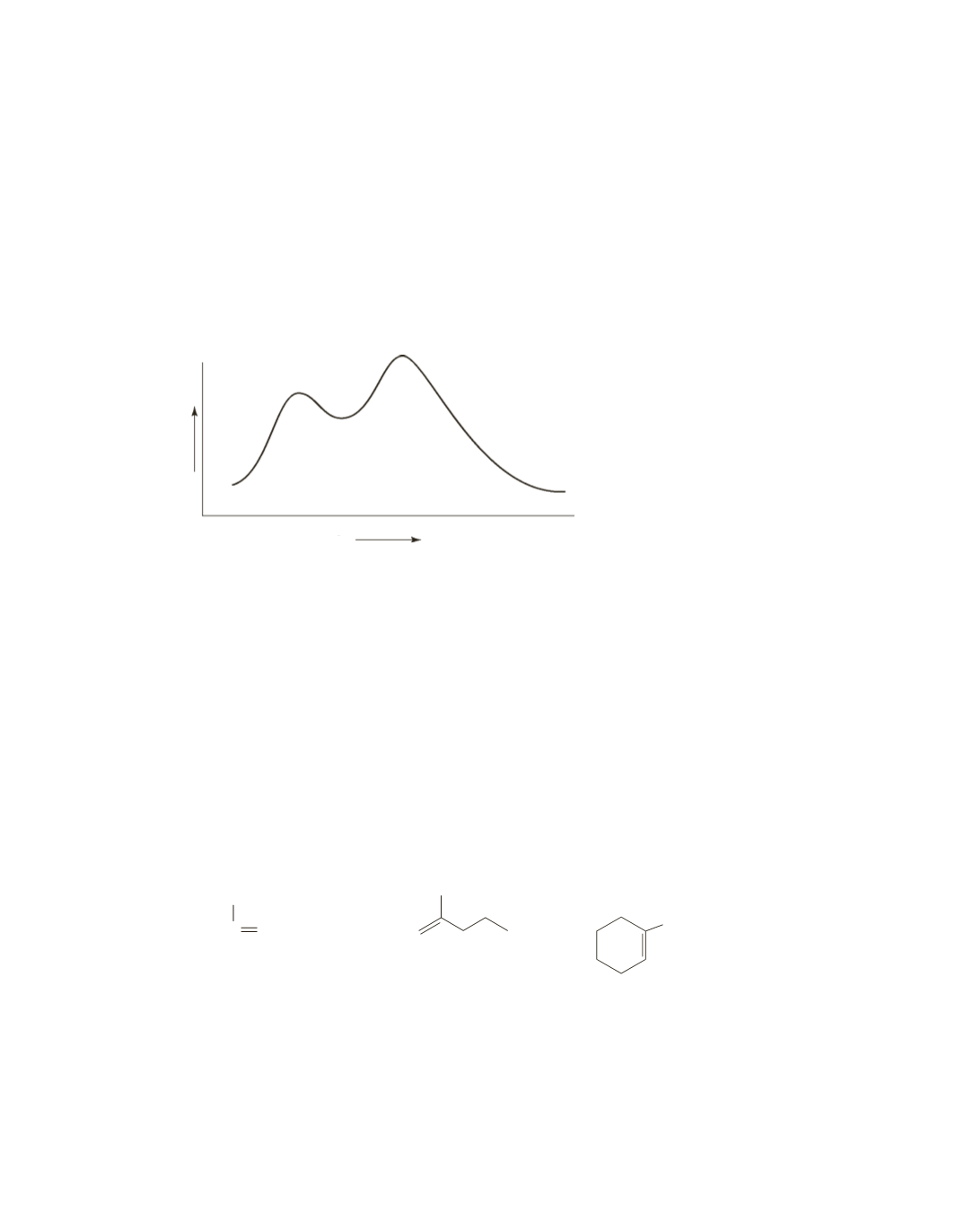
37.
Progress of the reaction
Free energy
A
B
C
a.
one (B)
b.
two
c.
the second step
1
k
2
2
, B to form C (In this particular diagram,
k
2
7
k
1
: if you had made the transition
state for the second step a lot higher, you could have had a diagram in which
k
1
7
k
2
.)
d.
the second step in the reverse direction
1
k
-
1
2
f.
B to C
e.
the second step in the reverse direction
1
k
-
1
2
, B to form A
g.
C to B
38.
A catalyst will change the energy difference between the reactants and the transition state, but it will not
change the energy difference between the reactants and the products.
∆
H
[
,
E
a
,
∆
S
[
,
∆
G
[
,
k
39.
a.
3,8-dibromo-4-nonene
b.
1
Z
2
-4-ethyl-3,7-dimethyl-3-octene
c.
1,5-dimethylcyclopentene
d.
3-ethyl-2-methyl-2-heptene
e.
4-methylcyclohexene
f.
4-ethyl-5-methylcyclohexene
40.
a.
CH C
3
CH
2
CH
3
CH
CH
3
b.
c.
CH
3
















