
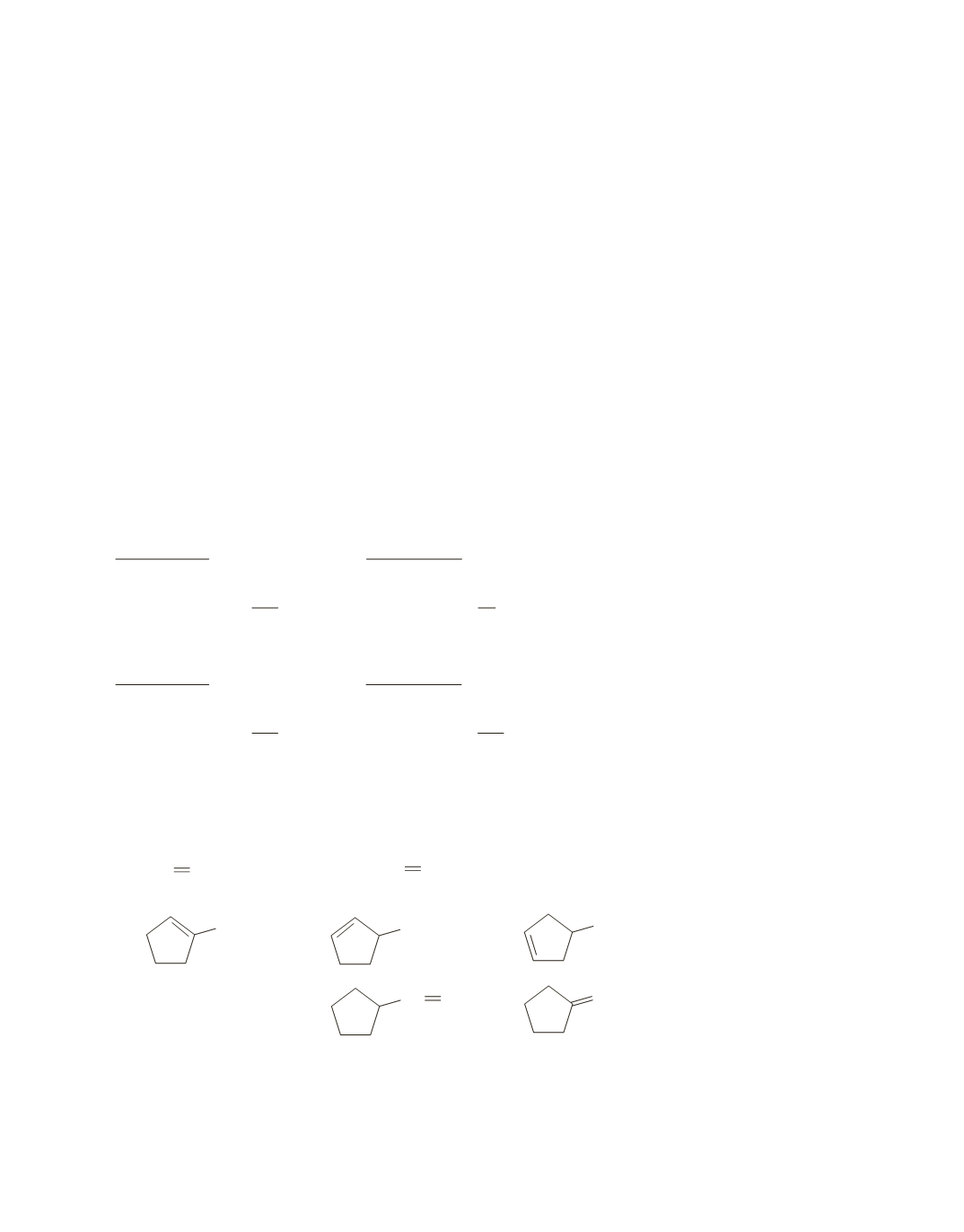
194 Chapter 5
Copyright © 2017 Pearson Education, Inc.
22.
a. 1.
∆
G
°
=
∆
H
°
-
T
∆
S
°
∆
G
°
=
-
RT
ln
K
eq
1
recall that
T
=
°
C
+
273
2
∆
G
°
=
-
1
1.986
*
10
-
3
2 1
303
2
ln
K
eq
∆
G
°
=
-
12
-
1
273
+
30
21
.01
2
-
15
=
-
0.60 ln
K
eq
∆
G
°
=
-
12
-
3
=
-
15 kcal
>
mol
ln
K
eq
=
25
K
eq
=
7.2
*
10
10
2.
∆
G
°
=
∆
H
°
-
T
∆
S
°
∆
G
°
=
-
1
1.986
*
10
-
3
2 1
423
2
ln
K
eq
∆
G
°
=
-
12
-
1
273
+
150
21
.01
2
-
16
=
-
0.84 ln
K
eq
∆
G
°
=
-
12
-
4
=
-
16 kcal
>
mol
ln
K
eq
=
19
K
eq
=
1.8
*
10
8
b.
For this reaction: the calculations show that increasing the temperature causes
∆
G
°
to be more negative.
c.
For this reaction: the calculations show that increasing the temperature causes
K
eq
to be smaller,
because ln
K
eq
=
-
∆
G
°
>
RT
.
23.
The value for the
p
bond of ethene (62 kcal/mol) is given in the text on page 43.
a.
bonds broken
bonds formed
p
bond of ethene 62
CH
3
CH
2
¬
H 101
H
¬
Cl
103
CH
3
CH
2
¬
Cl 85
165 kcal
>
mol
186 kcal
>
mol
∆
H
°
=
165
-
186
=
-
21 kcal
>
mol
b.
bonds broken
bonds formed
p
bond of ethene 62
CH
3
CH
2
¬
H 101
H
¬
H
104
CH
3
CH
2
¬
H 101
166 kcal
>
mol
202 kcal
>
mol
∆
H
°
=
166
-
202
=
-
36 kcal
>
mol
c.
Both are exothermic, because they both have a negative
∆
H
°
value.
d.
The
∆
H
°
values of both reactions are sufficiently negative to allow you to expect that they will be
exergonic as well.
24.
a.
b.
CH
2
CH
3
CHCH
2
CH
2
CH
3
CH
3
CH CHCH
2
CH
3
or
CH
2
CH
3
CH
2
CH
3
CH CH
2
CHCH
3
or
or
or
or
CH
2
25.
a.
three alkenes: 1-butene,
cis
-2-butene,
trans
-2-butene
b.
four alkenes: 3-methyl-1-pentene, (
E
)-3-methyl-2-pentene, (
Z
)-3-methyl-2-pentene, 2-ethyl-1-butene
c.
five alkenes: 1-hexene,
cis
-2-hexene,
trans
-2-hexene,
cis
-3-hexene,
trans
-3-hexene
















