
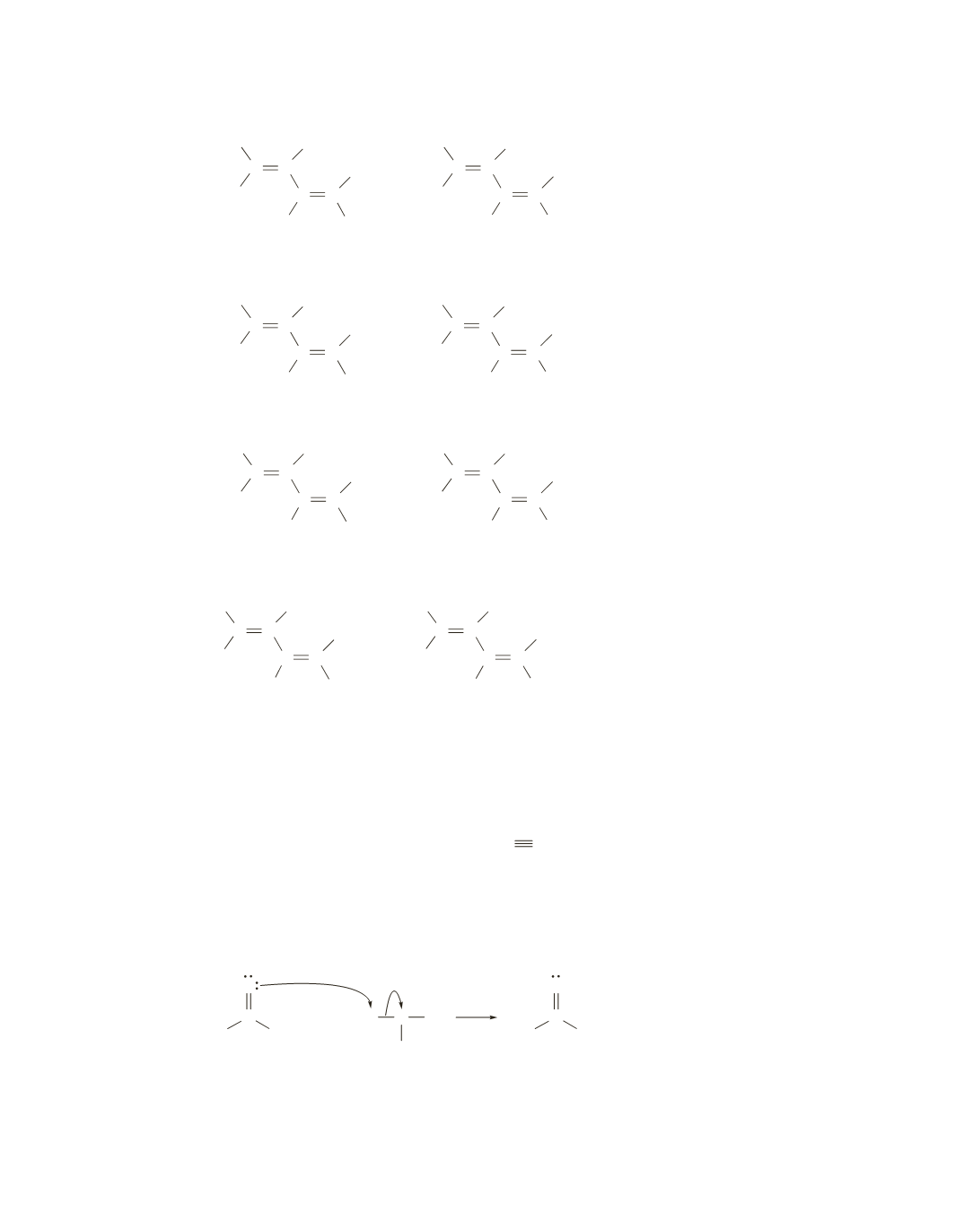
Chapter 5 191
Copyright © 2017 Pearson Education, Inc.
12.
a.
b.
C C
C
H
H CH
3
C
H
C C
C
H
H H
C
CH
3
H
C C
C
H
H CH
2
CH
3
C
H
H
C C
C
H
H H
C
CH
2
CH
3
C
H
C
C
H
H CH
2
CH
3
C
H
C
H
C
C
H
H H
C
CH
2
CH
3
c.
H
C
H
C
C
H
H CH
3
C
H
H
C
H
C
C
H
H H
C
CH
3
(
E
)-2-methyl-2,4-hexadiene (
Z
)-2-methyl-2,4-hexadiene
(2
E
,4
E
)-2,4-heptadiene
(2
E
,4
Z
)-2,4-heptadiene
(2
Z
,4
E
)-2,4-heptadiene
(2
Z
,4
Z
)-2,4-heptadiene
(
E
)-1,3-pentadiene
(
Z
)-1,3-pentadiene
CH
3
CH
3
CH
3
CH
3
CH
3
CH
3
CH
3
CH
3
b
has four stereoisomers because each double bond can have either the
E
or the
Z
configuration.
a
and
c
have only two stereoisomers because, in each case, there are two identical substituents bonded to
one of the
sp
2
carbons, so only one of the double bonds can have either the
E
or the
Z
configuration.
13.
nucleophiles:
H
−
3
CH O
−
3
CH C CH
3
NH
electrophiles:
+
3
3
CH CHCH
14.
a.
AlCl
3
is the electrophile, and NH
3
is the nucleophile.
b.
The H
d
+
of HBr is the electrophile, and HO
-
is the nucleophile.
15.
a.
nucleophile
electrophile
C
O
CH
3
OH
H H
H
2
O
O
H
+
+
+
C
CH
3
OH
+
OH
















