
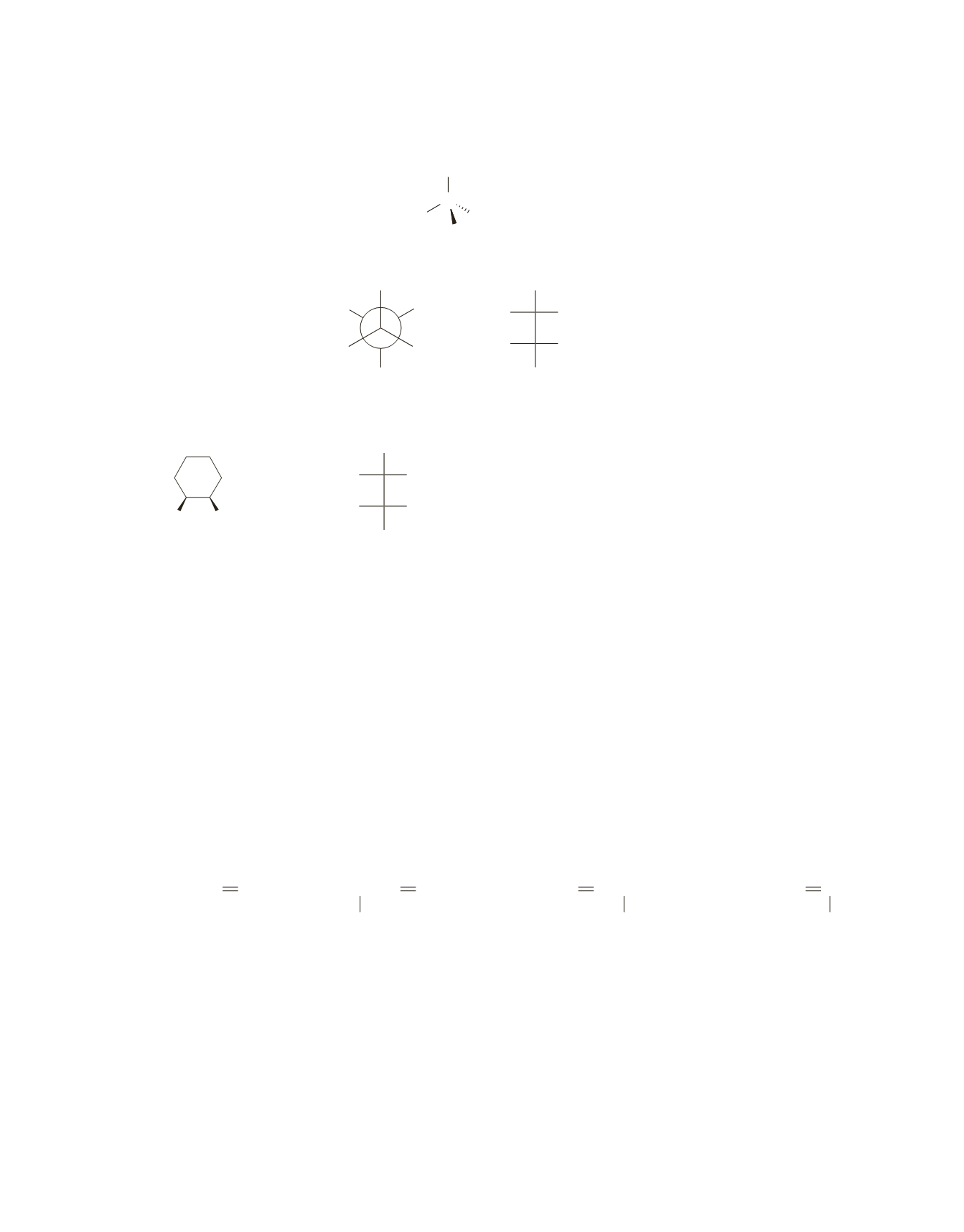
186 Chapter 4
Copyright © 2017 Pearson Education, Inc.
b.
Put the remaining groups on the structure so it represents (
R
)-2-butanol.
C
OH
H
c.
Put the remaining groups on the Fischer projection so it represents the Newman projection shown.
CH
3
CH
2
CH
3
CH
2
CH
3
HO
H
CH
3
Cl
H
d.
Draw a diastereomer for each of the following:
Cl
Cl
CH
3
CH
3
Br
H
OH
H
1.
2.
14.
Indicate whether each of the following statements is true or false:
a.
Diastereomers have the same melting points.
T F
b.
3-Chloro-2,3-dimethylpentane has two asymmetric centers.
T F
c.
Meso compounds do not rotate the plane of polarization of
plane-polarized light.
T F
d.
2,3-Dichloropentane has a stereoisomer that is a meso compound.
T F
e.
All chiral compounds with the
R
configuration are dextrorotatory.
T F
f.
A compound with three asymmetric centers can have a maximum
of nine stereoisomers.
T F
15.
Which of the following have cis–trans isomers?
CH
3
CH
2
CH
2
CH CH
2
CH
3
CH
3
CH
2
CH
2
CH CCH
3
CH
3
CH
3
CH
2
CHCH CHCH
3
Br
CH
3
CH
2
CH CHCHCH
3
















