
196 Chapter 5
Copyright © 2017 Pearson Education, Inc.
31.
a.
Solved in the text.
b.
Decreasing the concentration of methyl chloride (by a factor of 10) decreases
the rate of the reaction
(by a factor of 10) to 1
*
10
-
8
M s
-
1
.
32.
The rate constant for a reaction can be increased by
decreasing
the stability of the reactant (increasing its
energy) or by
increasing
the stability of the transition state (decreasing its energy).
33.
Taking the logarithm of both sides of the Arrhenius equation gives the following equation (where
k
is the
rate constant), which we can use to answer the questions:
ln
k
=
ln
A
-
E
a
RT
a.
Increasing the experimental activation energy
1
E
a
2
decreases the rate constant of a reaction (causes the
reaction to be slower).
b.
Increasing the temperature
1
T
2
increases the rate constant of a reaction (causes the reaction to go
faster).
34.
a.
The first stated reaction has the greater equilibrium constant:
K
eq
=
1
*
10
-
3
1
*
10
-
5
=
1
*
10
2
K
eq
=
1
*
10
-
2
1
*
10
-
3
=
10
b.
Because both reactions start with the same concentration, the first stated reaction will form the most
product when the reactions have reached equilibrium, because it has the greater equilibrium constant.
35.
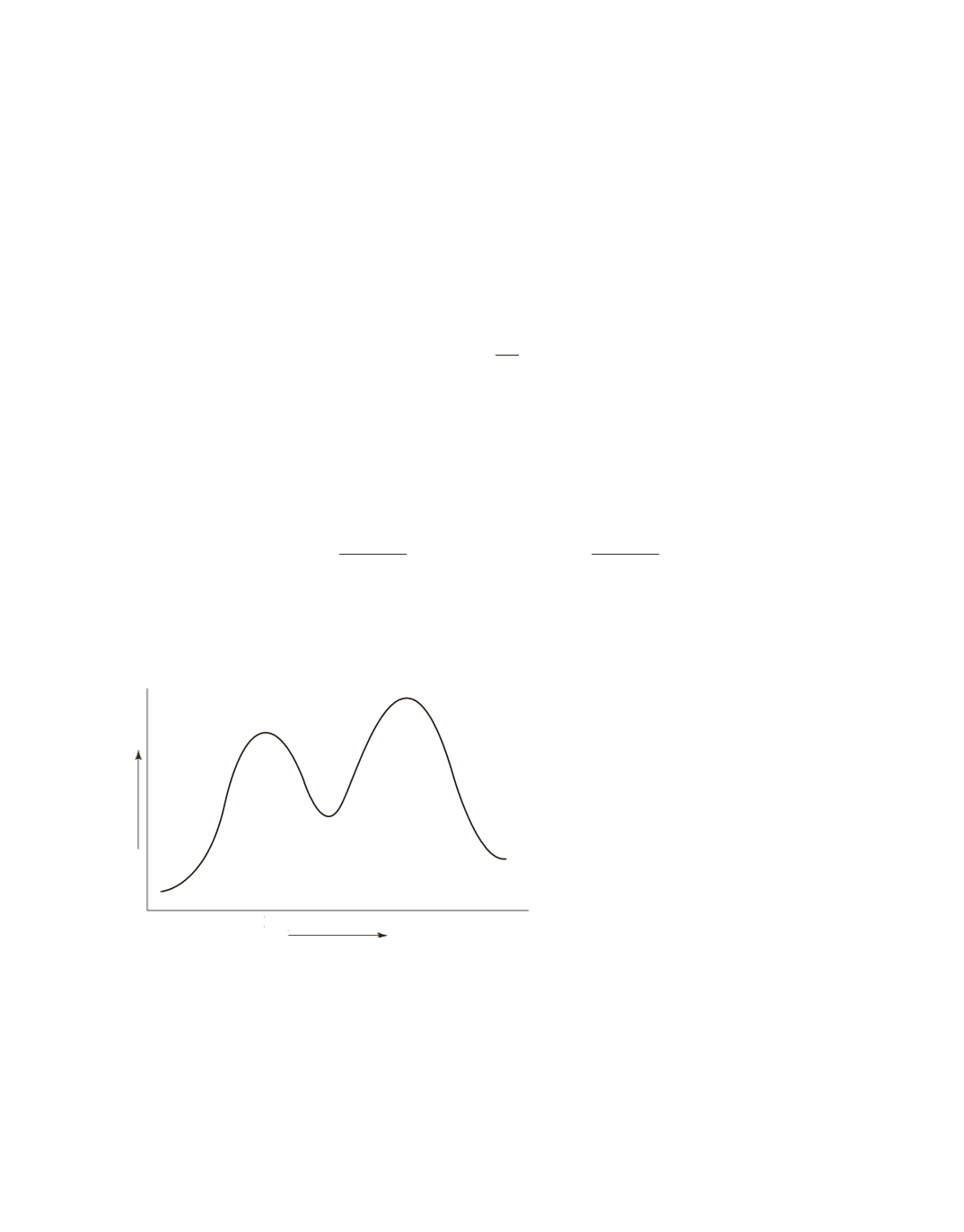
D
Progress of the reaction
A
= reactant(s)
B
= rst transition state
C
= intermediate
D
= second transition state
E
= product(s)
Free energy
A
B
C
E
















