
Chapter 5 195
Copyright © 2017 Pearson Education, Inc.
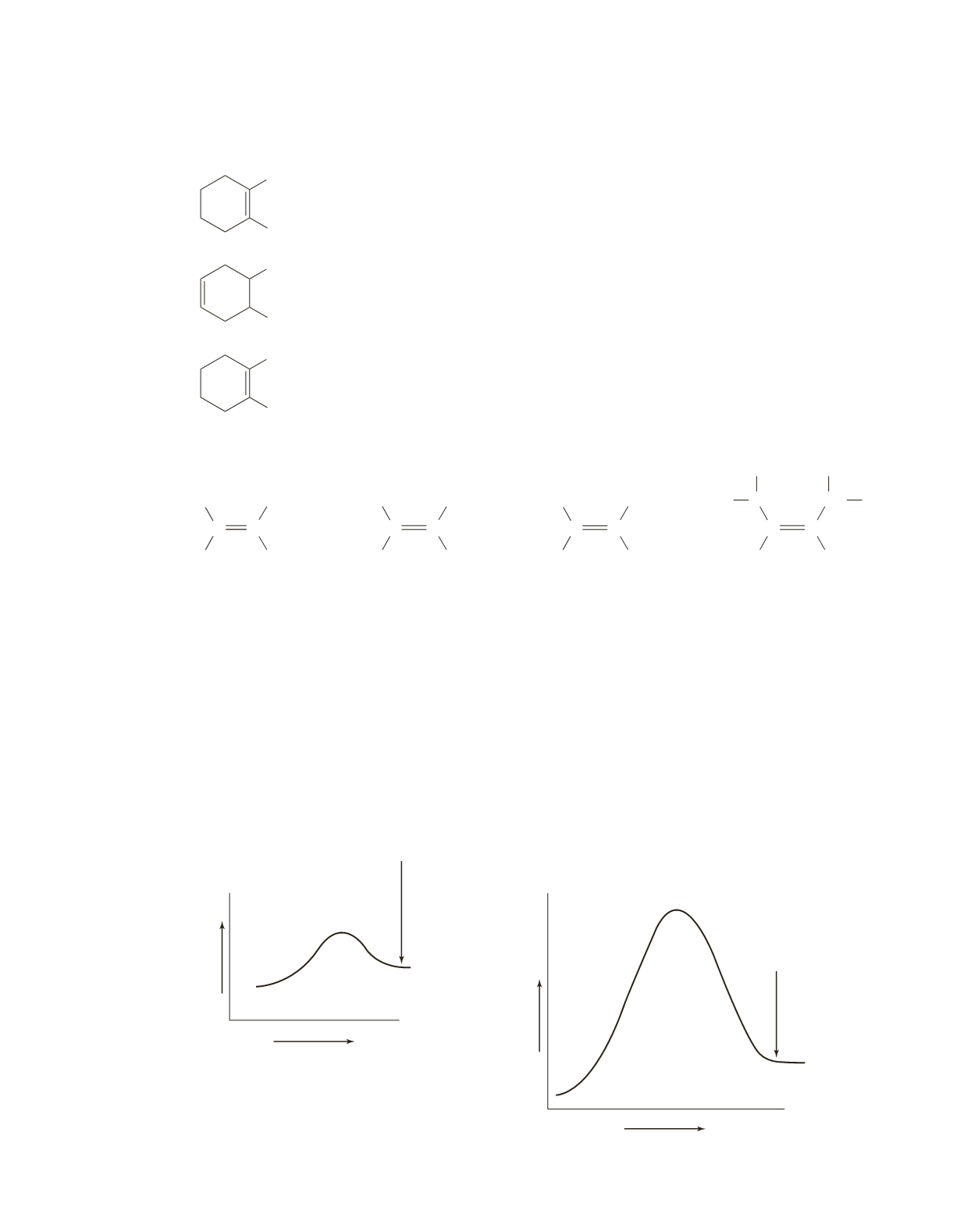
26.
Because alkene
A
has the smaller heat of hydrogenation, it is more stable.
27.
a.
CH
2
CH
3
CH
2
CH
3
This alkene is the most stable because it has the greatest
number of alkyl substituents bonded to the
sp
2
carbons.
b.
CH
2
CH
3
CH
2
CH
3
This alkene is the least stable because it has the fewest
number of alkyl substituents bonded to the
sp
2
carbons.
c.
CH
2
CH
3
CH
2
CH
3
This alkene has the smallest heat of hydrogenation
because it is the most stable of the three alkenes.
28.
C C
CH
3
CH
2
H
CH
2
CH
3
H
2 trans alkyl substituents
>
2 cis alkyl substituents
>
C C
H
CH CH
H
2 cis alkyl substituents
that cause greater steric strain
than those in
cis
-3-hexene
CH
3
CH
3
C C
H
CH
3
CH
2
CH
2
CH
3
H
>
C C
CH
2
CH
3
CH
3
CH
3
CH
2
4 alkyl substituents
CH
3
CH
3
CH
3
29.
a. a
and
b
, because the product is more stable than the reactant.
b. b
is the most kinetically stable product, because it has the smallest rate constant
1
greatest
∆
G
[
2
lead-
ing from the product to the transition state.
c. c
is the least kinetically stable product, because it has the largest rate constant
1
smallest
∆
G
[
2
leading
from the product to the transition state.
30.
a.
A thermodynamically
unstable
product is less stable than the reactant.
A kinetically
unstable
product has a large rate constant
1
small
∆
G
[
2
for the reverse reaction.
b.
A kinetically
stable
product has a small rate constant
1
large
∆
G
[
2
for the reverse reaction.
A thermodynamically
unstable product
A kinetically
unstable
product
Free
energy
Free
energy
Progress of the reaction
Progress of the reaction
A thermodynamically
unstable product
A kinetically stable
product
a.
b.
















