
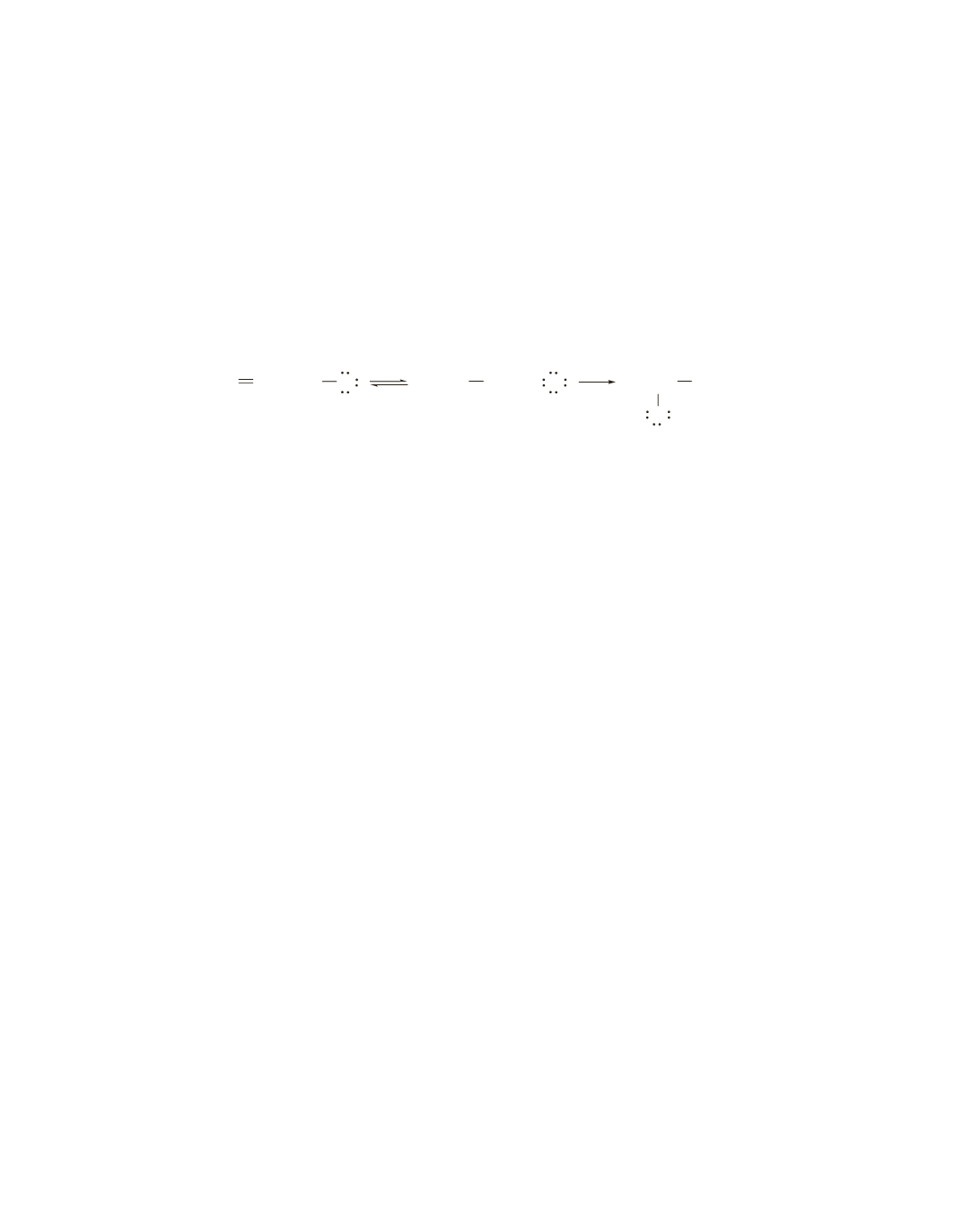
206 Chapter 5
Copyright © 2017 Pearson Education, Inc.
5.
The addition of H
2
in the presence of Pd/C to alkenes
A
and
B
results in the formation of the same alkane.
The addition of H
2
to alkene
A
has a heat of hydrogenation
1
-
∆
H
°
2
of 29.7 kcal
>
mol, whereas the addition
of H
2
to alkene
B
has a heat of hydrogenation of 27.3 kcal/mol. Which is the more stable alkene,
A
or
B
?
6.
Draw structures for each of the following:
a.
allyl alcohol
b.
3-methylcyclohexene
c.
cis
-3-heptene
d.
vinyl bromide
7.
What is the total number of
p
bonds and rings in a hydrocarbon with a molecular formula of C
8
H
8
?
8.
Using curved arrows, show the movement of electrons in the following mechanism:
CH
3
CH CH
2
H Cl
CH
3
CH CH
3
+
CH
3
CH
Cl
CH
3
+
Cl
+
−
9.
A favorable (negative)
∆
G
°
is given by:
a positive or negative
∆
H
°
, a positive or negative
∆
S
°
, a high or low temperature.
10.
Which of the following has a more favorable equilibrium constant (that is, which reaction favors products
more)?
a.
A reaction with a
∆
H
°
of 4 kcal/mol or a reaction with a
∆
H
°
of 7 kcal/mol? (Assume a constant
∆
S
°
value.)
b.
A reaction with a positive
∆
S
°
value that takes place at 25
°
C or the same reaction that takes place at
35
°
C?
c.
A reaction in which two reactants form one product or a reaction in which one reactant forms two
products? (Assume a constant
∆
H
°
value.)
11.
Draw a reaction coordinate diagram for a one-step reaction with a product that is thermodynamically
unstable but kinetically stable.
















