
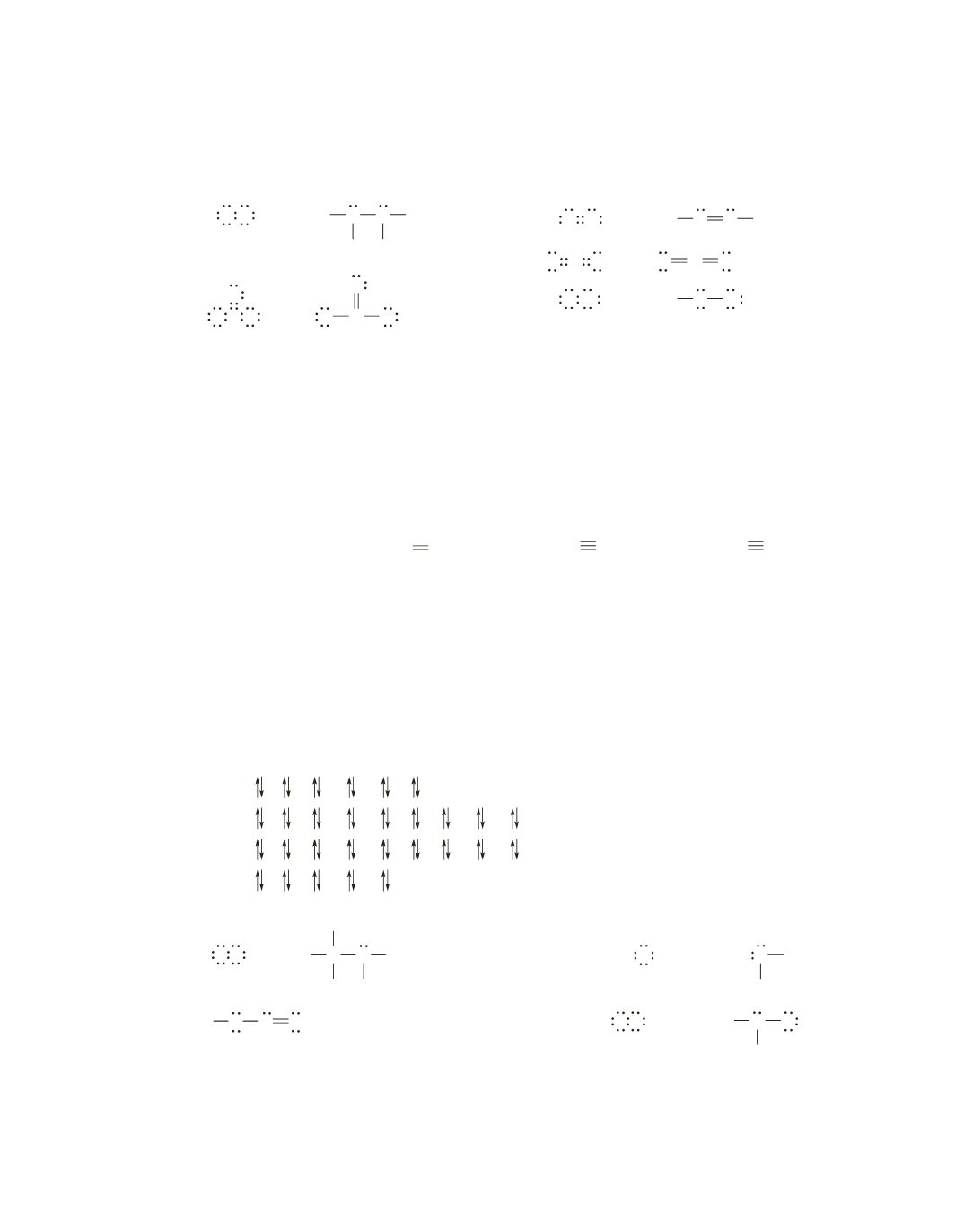
Chapter 1
85
Copyright © 2017 Pearson Education, Inc.
48.
The atom with the greater electronegativity will decrease the electron flow toward the electronegative F
atom, giving the compound a smaller dipole moment. Since
CH
3
F
has a smaller dipole moment than
CD
3
F,
we know that hydrogen is more electronegative than deuterium.
49.
a.
or
NH
H
N H
H
N
H
H N
H
H
b.
C
O C O
−
O O
O
O
or
−
−
−
c.
or
NH N H
NH N H
d.
O
C O
O C
O
or
e.
or
OH
Cl
OH Cl
50.
a.
CH
3
NH
2
,
CH
3
F
,
CH
3
OH
b.
CH
3
F
51.
If the central atom is
sp
3
hybridized, the bond angle will depend on the number of lone pairs it has: no lone
pairs
=
109.5°; one lone pair
=
107.3°; two lone pairs
=
104.5°.
a.
sp
3
,
107.3
°
b.
sp
2
,
120
°
c.
sp
3
,
107.3
°
d.
sp
2
,
120
°
e.
sp
3
,
109.5
°
f.
sp
2
,
120
°
g.
sp
, 180
°
h.
sp
3
,
109.5
°
i.
sp
3
,
107.3
°
j.
sp
2
,
120
°
52.
a.
CH
3
CH
2
CH
3
b.
3
2
CH CH CH
c.
CH
3
C CCH
3
or
CH
3
CH
2
C CH
53.
The hybridization of the central atom determines the bond angle. If the hybridization is
sp
3
,
the number of
lone pairs on the central atom determines the bond angle.
a.
109.5
°
b.
104.5
°
*
c.
107.3
°
d.
107.3
°
*104.5
°
is the correct prediction based on the bond angle in water.
However, the bond angle is actually somewhat larger
1
108.2
°
2
because the bond opens up to minimize the
interaction between the electron cloud of the relatively bulky
CH
3
group.
54.
1
s
2
s
2
p
x
2
p
y
2
p
z
3
s
3
p
x
3
p
y
3
p
z
Mg
Ca
2
+
Ar
Mg
2
+
a.
b.
c.
d.
1
s
2
2
s
2
2
p
6
3
s
2
1
s
2
2
s
2
2
p
6
3
s
2
3
p
6
1
s
2
2
s
2
2
p
6
3
s
2
3
p
6
1
s
2
2
s
2
2
p
6
55.
a.
N
C N H
H C H
H
H
or
HH
H
H H
b.
O N O
H
c.
or
N
H
Na N H
H
+ –
Na
+ –
H
d.
O
N O
H N
−
H
or
H
H
−
















